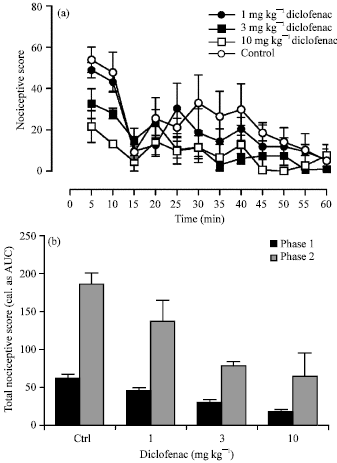
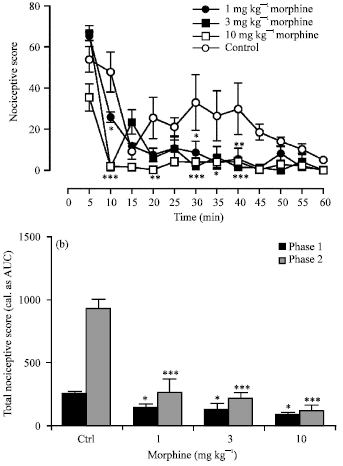
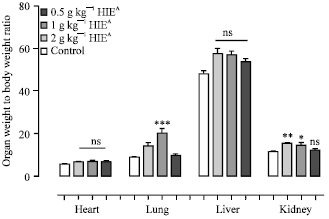
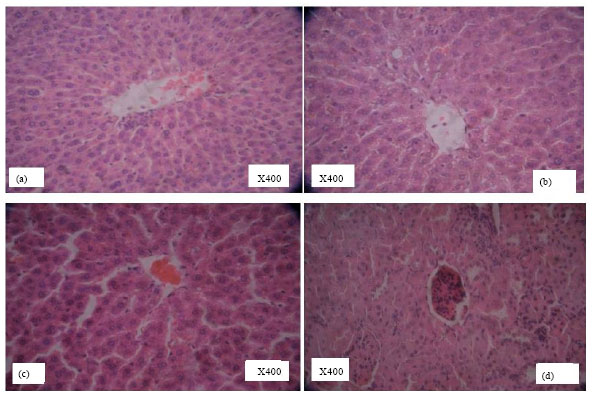
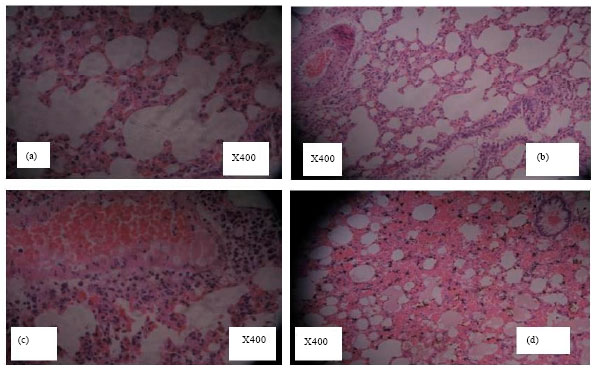
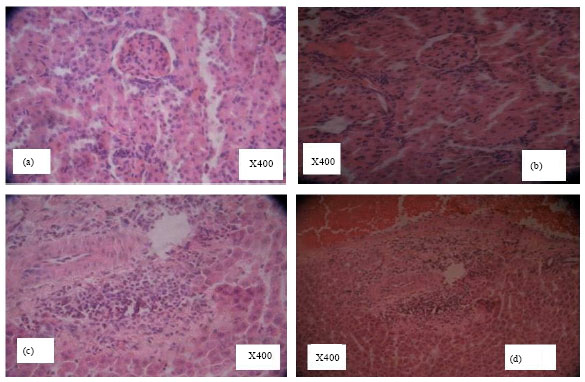
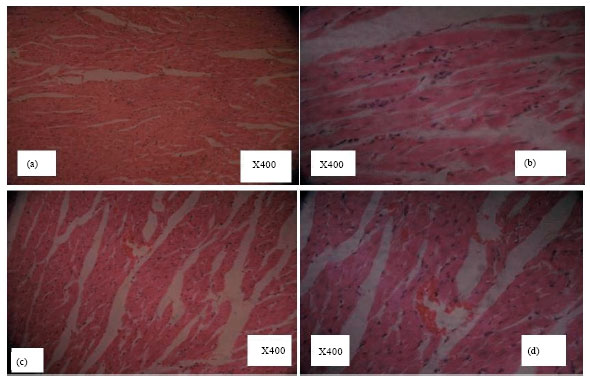
Research Article
Analgesic Activity and Safety Assessment of Heliotropium indicum Linn. (Boraginaceae) in Rodents
Department of Medical Laboratory Technology, School of Physical Sciences, University of Cape Coast, Cape-Coast, Ghana
G.A. Koffuor
Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
P. Amoateng
Department of Pharmacology and Toxicology, School of Pharmacy, College of Health Sciences, University of Ghana, Legon-Accra, Ghana
E.O. Ameyaw
Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
A.K. Abaitey
Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana