Research Article
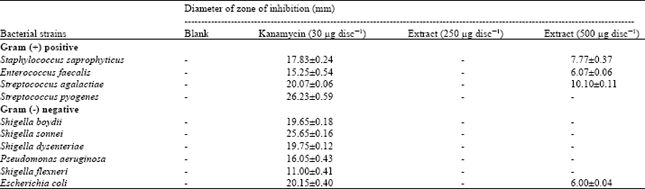
Phytochemical Investigation and Evaluation of Antibacterial and Antioxidant Potentials of Asparagus racemosus
Pharmacy Discipline, Khulna University, Khulna, 9208, Bangladesh
S.K. Biswas
Department of Pharmacy, BGC Trust University Bangladesh, Chittagong, Bangladesh
A. Chowdhury
Department of Pharmacy, BGC Trust University Bangladesh, Chittagong, Bangladesh
S.Z. Raihan
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh
M.A. Akbar
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh
M.A. Muhit
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh
R. Mowla
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh