Research Article
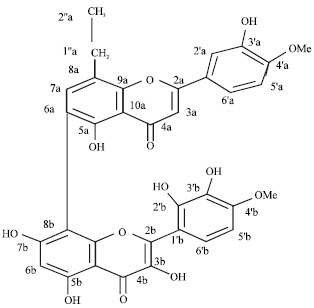
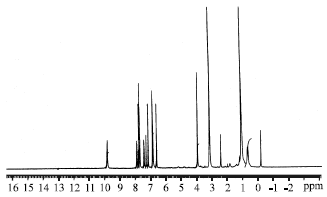
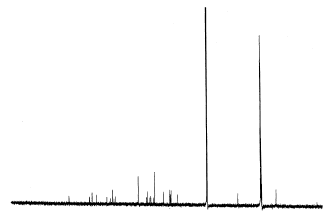
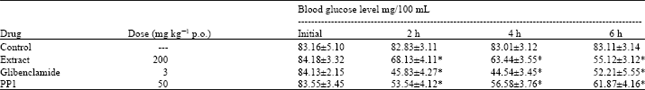
Isolation and Hypoglycemic Activity of a Novel Pongamiaflavonylflavonol from Pongamia pinnata Pods
Department of Pharmaceutical Sciences, H.N.B. Garhwal University Srinagar (Garhwal), India
M. Semalty
Department of Pharmaceutical Sciences, H.N.B. Garhwal University Srinagar (Garhwal), India
P. Kumar
Department of Pharmaceutical Sciences, H.N.B. Garhwal University Srinagar (Garhwal), India
S.R. Mir
Faculty of Pharmacy, Jamia Hamdard, New Delhi, India
M. Ali
Faculty of Pharmacy, Jamia Hamdard, New Delhi, India
S. Amin
Faculty of Pharmacy, Jamia Hamdard, New Delhi, India