Review Article
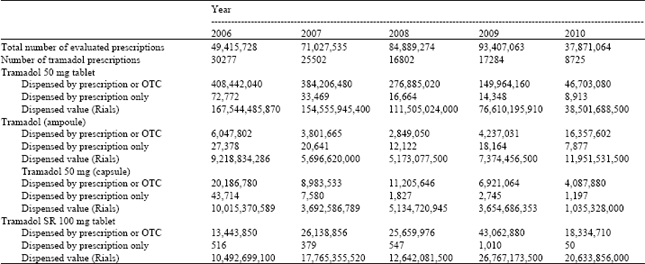
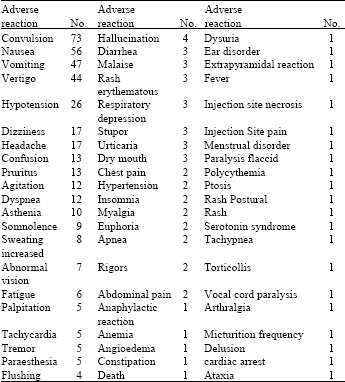
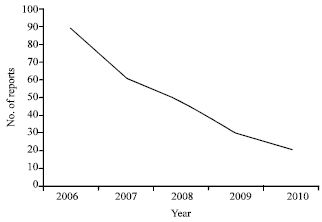
Pattern of use and Adverse Drug Reactions of Tramadol; A Review of 336, 610, 664 Insured Prescriptions During 5 Years
Department of Pharmacoeconomics and Pharmaceutical Management, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
G. Shalviri
Iranian Adverse Drug Reaction Monitoring Center, Food and Drug Organization, Ministry of Health and Medical Education, Tehran, Iran
M. Abdollahi
Department of Pharmacoeconomics and Pharmaceutical Management, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran