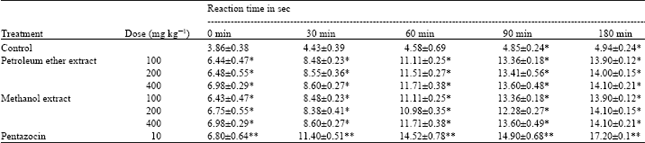
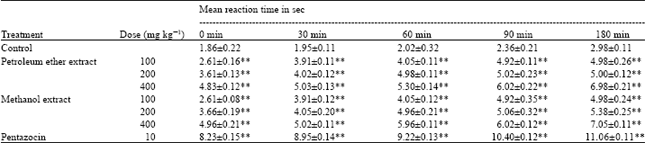
Research Article
Analgesic Activity of Abelmoschus manihot Extracts
Department of Pharmaceutical Chemistry, R.C. Patel Institute of Pharmaceutical Education and Research, Near Karwand Naka, Shirpur, Dist. Dhule, Maharashtra-425 405, India
A. Todarwal Amol
Department of Pharmaceutical Chemistry, R.C. Patel Institute of Pharmaceutical Education and Research, Near Karwand Naka, Shirpur, Dist. Dhule, Maharashtra-425 405, India
B. Bari Sanjay
Department of Pharmaceutical Chemistry, R.C. Patel Institute of Pharmaceutical Education and Research, Near Karwand Naka, Shirpur, Dist. Dhule, Maharashtra-425 405, India
J. Surana Sanjay
Department of Pharmaceutical Chemistry, R.C. Patel Institute of Pharmaceutical Education and Research, Near Karwand Naka, Shirpur, Dist. Dhule, Maharashtra-425 405, India