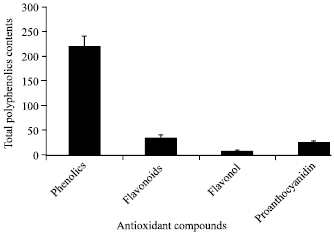
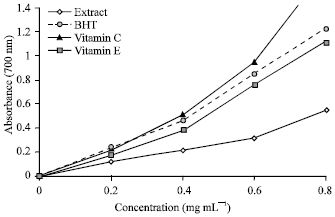
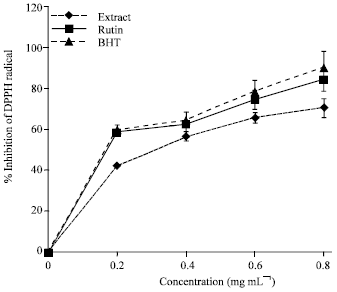
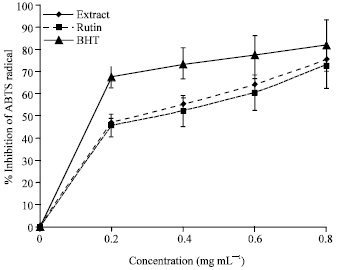
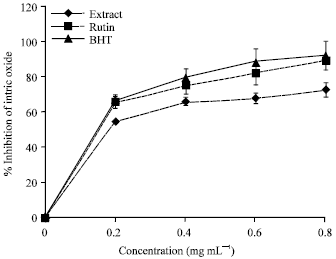
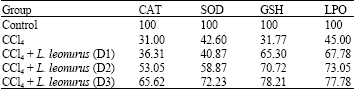
Research Article
In vitro and in vivo Antioxidant Activity of Aqueous Leaves Extract of Leonotis leonurus (L.) R. Br.
School of Biological Sciences, University of Fort Hare, Alice 5700, South Africa
A. J. Afolayan
School of Biological Sciences, University of Fort Hare, Alice 5700, South Africa