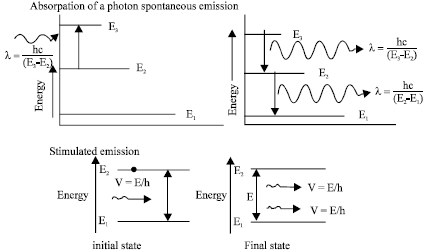
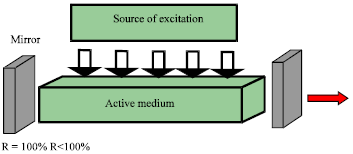
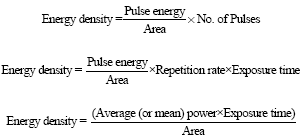
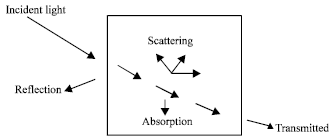
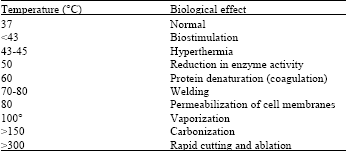
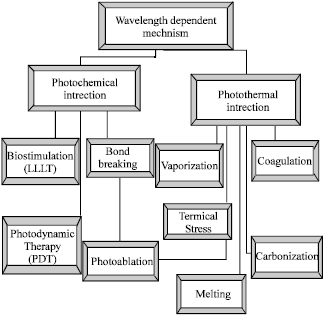
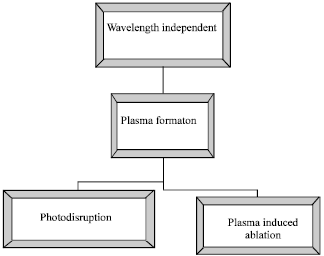
Review Article
An Overview of Laser Principle, Laser-Tissue Interaction Mechanisms and Laser Safety Precautions for Medical Laser Users
Department of Medical Engineering, College of Engineering, Al-Nahrain University, Baghdad, Iraq
Sarah T. Abdul Qader
Department of Paediatric and Preventive Dentistry, College of Dentistry, University of Baghdad, Baghdad, Iraq
A. A. Zaidan
Faculty of Engineering, Multimedia University, 63100, Selangor Darul Ehsan Cyberjaya, Malaysia
B. B. Zaidan
Faculty of Engineering, Multimedia University, 63100, Selangor Darul Ehsan Cyberjaya, Malaysia
A. W. Naji
Faculty of Engineering, International Islamic University Malaysia (IIUM), 53100 Gombak, Kuala Lumpur, Malaysia
Ibraheem T. Abdul Qader
Faculty of Engineering, International Islamic University Malaysia (IIUM), 53100 Gombak, Kuala Lumpur, Malaysia