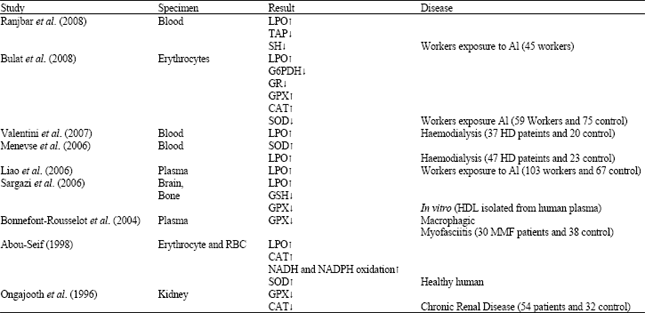
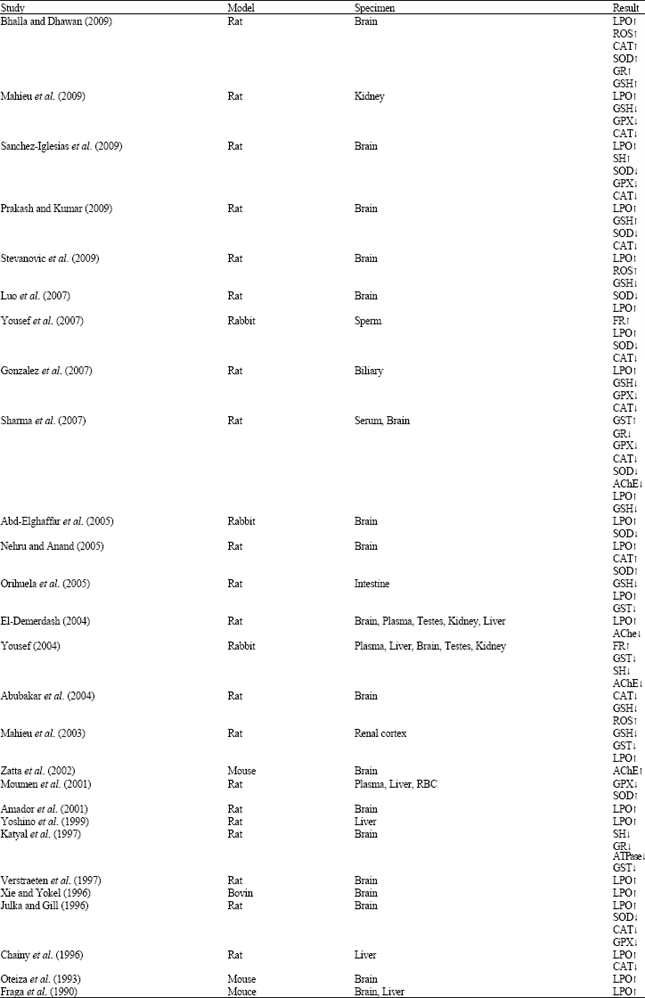
Review Article
A Systematic Review on Oxidant/Antioxidant Imbalance in Aluminium Toxicity
Faculty of Pharmacy and Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran 1417614411, Iran
Mohammad Abdollahi
Faculty of Pharmacy and Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran 1417614411, Iran