Research Article
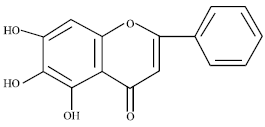
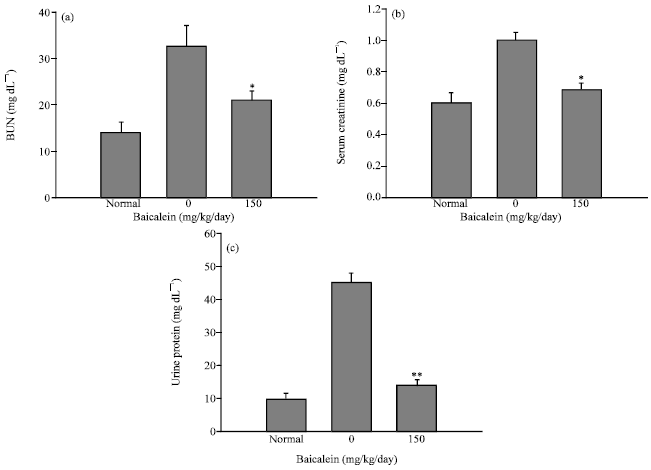
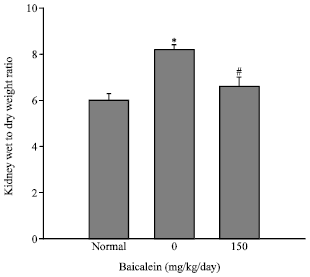
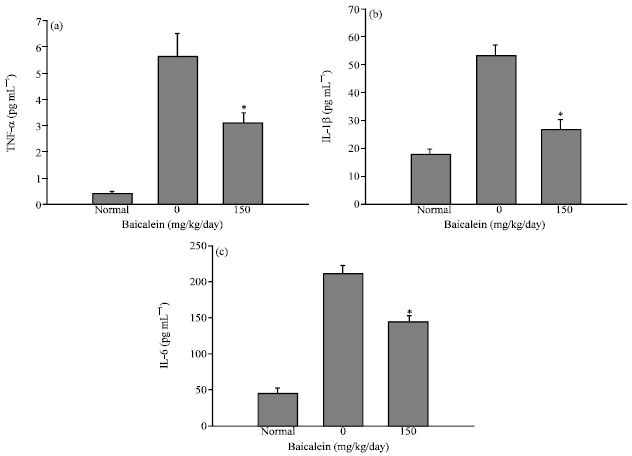
Protective Effect of Baicalein Extracted from Scutellaria baicalensis against Lipopolysaccharide-Induced Glomerulonephritis in Mice
Department of Pathology and Laboratory Medicine, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan
Hui-Fen Chiu
Department of Pharmacology, College of Medicine, Kaoshuing Medical University, Kaohsiung, Taiwan
Jyh-Seng Wang
Department of Pathology and Laboratory Medicine, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan
Jenn-Kuen Lee
Department of Pathology and Laboratory Medicine, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan
Tz-Chong Chou
Department of Physiology, National Defense Medical Center, Taipei, Taiwan