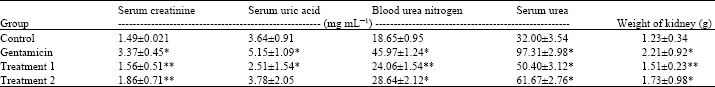
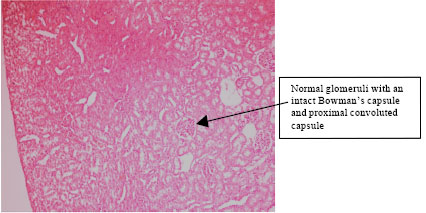
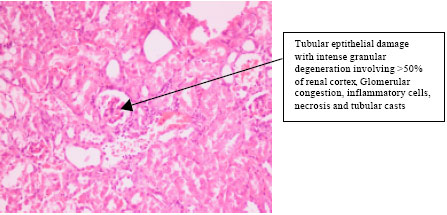
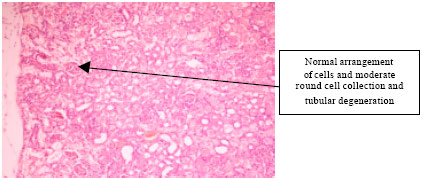
Research Article
Protective Effect of Zingiber officinale on Gentamicin-Induced Nephrotoxicity in Rats
Department of Pharmacology, Malla Reddy College of Pharmacy, Dhulapally (via Hakimpet), Maisammaguda, Secunderabad, 500014, Andhra Pradesh, India
M. Sudhakar
Department of Pharmacology, Malla Reddy College of Pharmacy, Dhulapally (via Hakimpet), Maisammaguda, Secunderabad, 500014, Andhra Pradesh, India