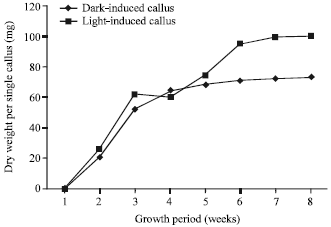
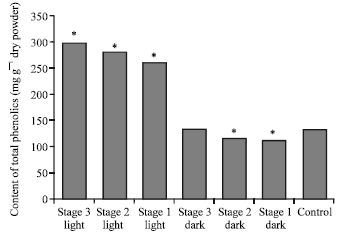
Research Article
Phytochemical Screening, Total Phenolics and Antioxidant and Antibacterial Activities of Callus from Brassica nigra L. Hypocotyl Explants
Department of Biology, Faculty of Science, Ibb University, Ibb, Yemen
A. M. Taj-Eldeen
Center of Genetic Resources, Faculty of Agriculture, Sana`a University, Sana a, Yemen
A. S. Al-Zubairi
Department of Biochemistry and Molecular Biology, Faculty of Medicine and Health Sciences, University of Sana`a, Yemen
A. S. Elhakimi
Center of Genetic Resources, Faculty of Agriculture, Sana`a University, Sana a, Yemen
A. R. Al-Dubaie
Department of Biology, Faculty of Science, Sana a University, Sana`a, Yemen