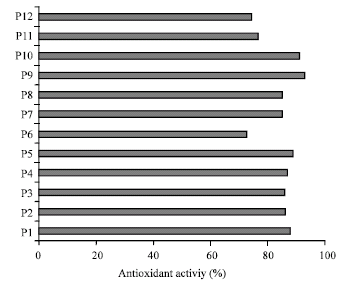
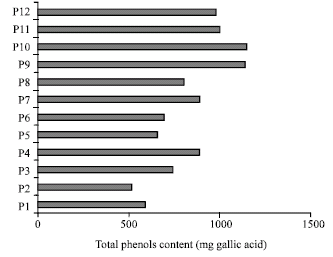
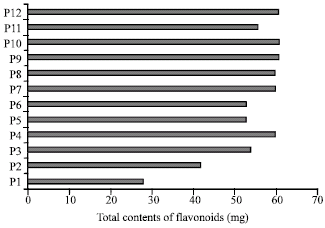
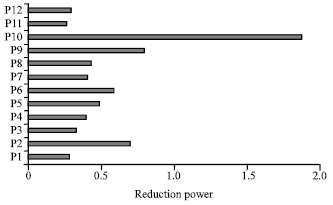
Research Article
The Obtaining of an Antioxidant Product Based on a Rosmarinus officinalis Freeze-Dried Extract
Applied Biochemistry and Biotechnology Center, Biotehnol, Bd. Maraoti No. 59, Sector 1, Bucharest, Romania
V. Adrian
Applied Biochemistry and Biotechnology Center, Biotehnol, Bd. Maraoti No. 59, Sector 1, Bucharest, Romania
Nita Sultana
National Institute of Chemical-Pharmaceutical Research-Development, ICCF Bucharest, Vitan Road No. 112, Sector 3, Bucharest, Romania