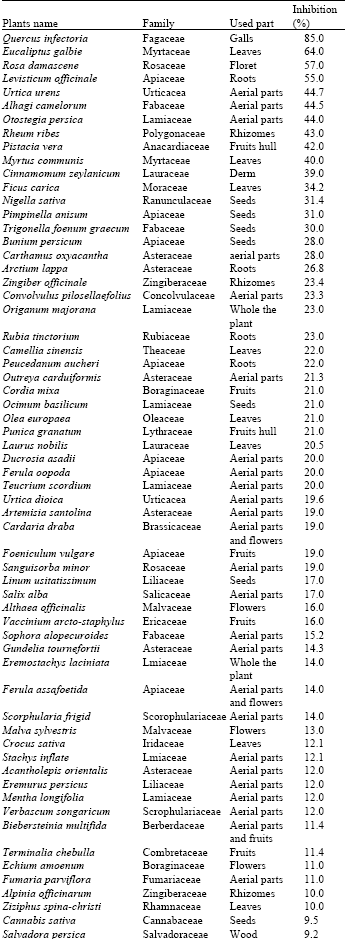
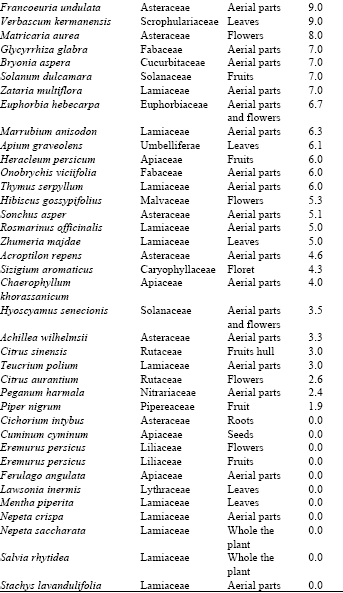
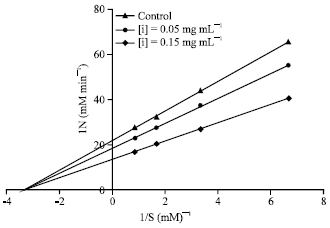
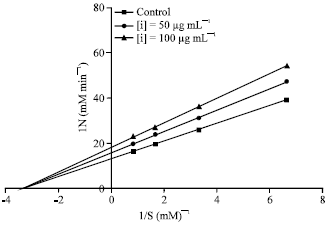
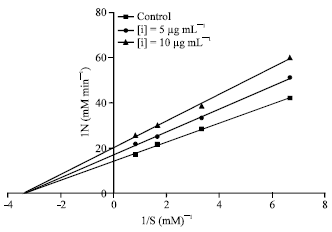
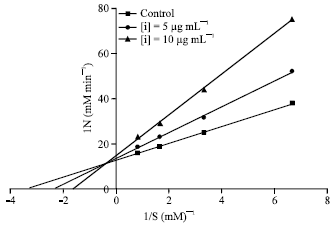
Research Article
Inhibitory Effect of Some Plant Extracts on Pancreatic Lipase
Department of Biochemistry, Medical School and Kerman Physiology Research Center,
B. Shahouzehi
Department of Biochemistry, Medical School and Kerman Physiology Research Center,
F. Sharifi-far
Department of Pharmacognosy, School of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran