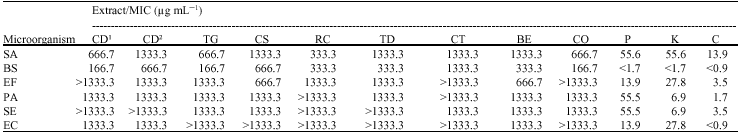
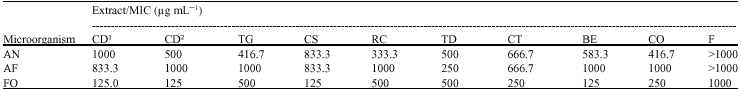
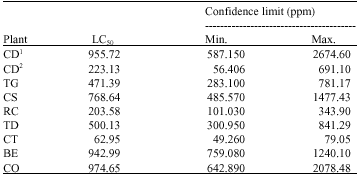
Research Article
Antibacterial, Antifungal and Toxicity of Rare Iranian Plants
Pharmaceutical Design and Bioinformatics Group, Biotechnology Research Center, Department of Biotechnology, Pasteur Institute, No. 69 Pasteur Avenue, 13164, Tehran, Iran
J. Hamedi
Microbial Biotechnology Laboratory, Department of Microbiology, School of Biology, College of Science, University of Tehran, P.O. Box 14155-6455, Tehran, Iran
S. Moradi
Pharmaceutical Design and Bioinformatics Group, Biotechnology Research Center, Department of Biotechnology, Pasteur Institute, No. 69 Pasteur Avenue, 13164, Tehran, Iran
F. Mohammadipanah
Microbial Biotechnology Laboratory, Department of Microbiology, School of Biology, College of Science, University of Tehran, P.O. Box 14155-6455, Tehran, Iran
S. Sardari
Pharmaceutical Design and Bioinformatics Group, Biotechnology Research Center, Department of Biotechnology, Pasteur Institute, No. 69 Pasteur Avenue, 13164, Tehran, Iran