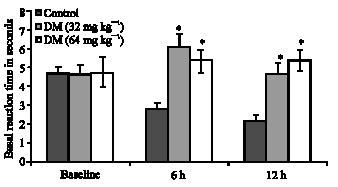
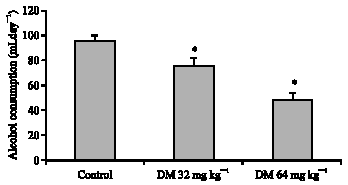
Research Article
Dextromethorphan Attenuates Ethanol Withdrawal Induced Hyperalgesia in Rats
Department of Pharmacology, K.S. Hegde Medical Academy, Mangalore 575 018, India
K.S. Karanth
Department of Pharmacology, K.S. Hegde Medical Academy, Mangalore 575 018, India