Research Article
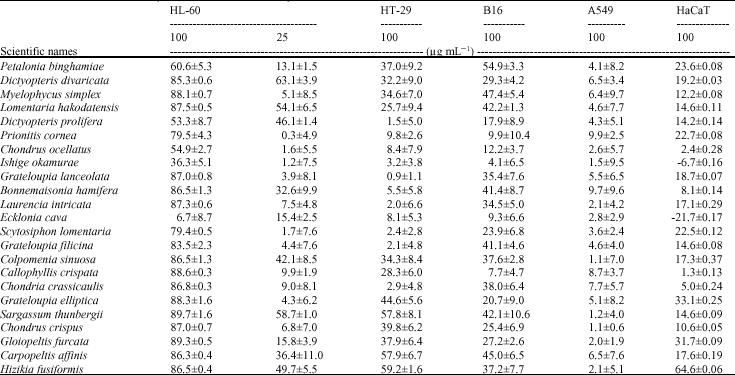
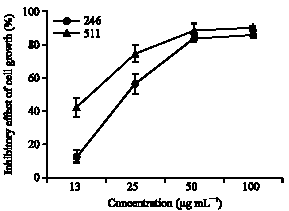
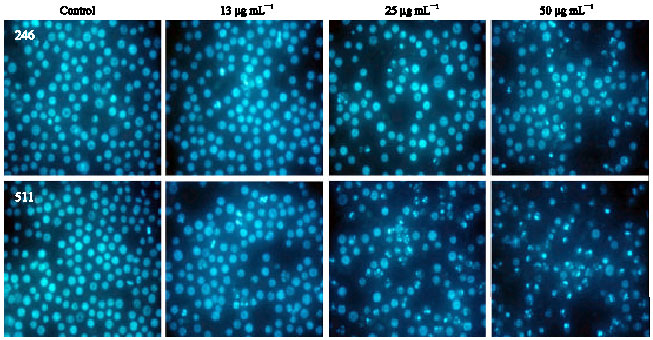
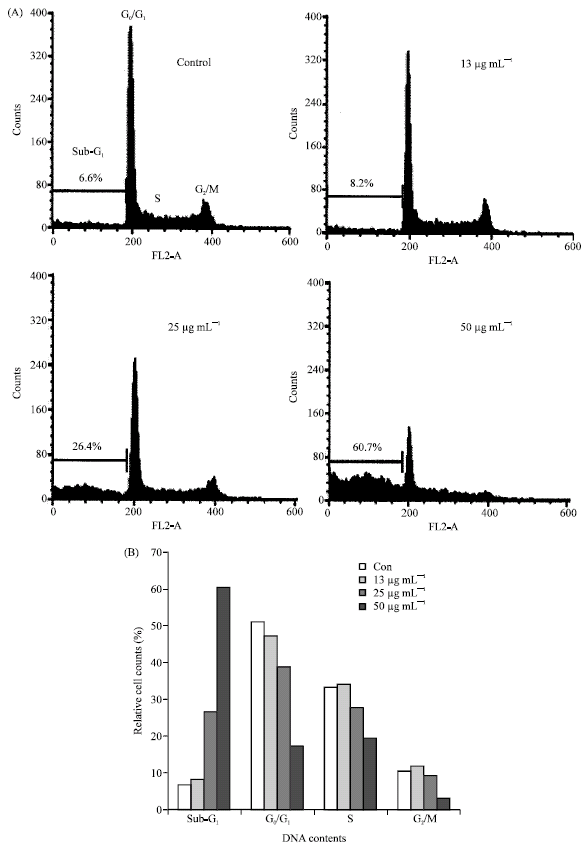
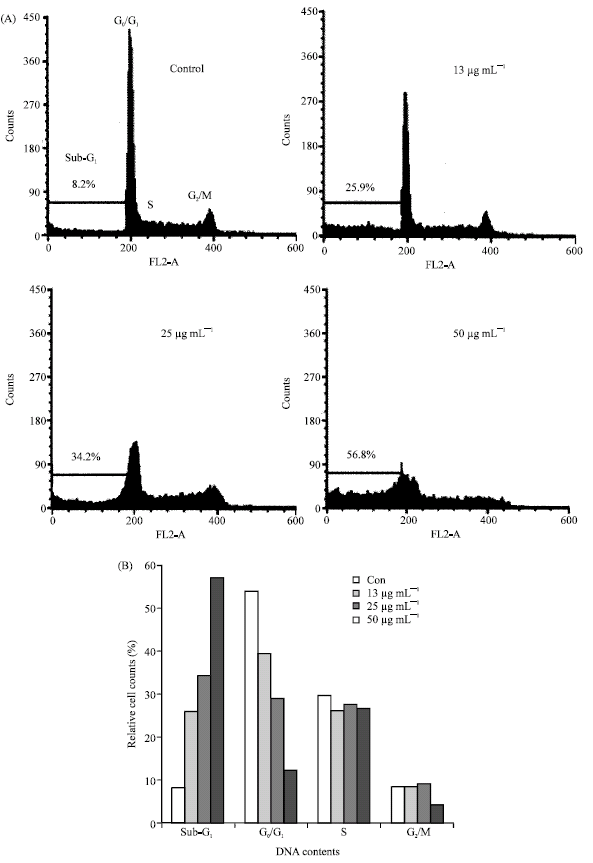
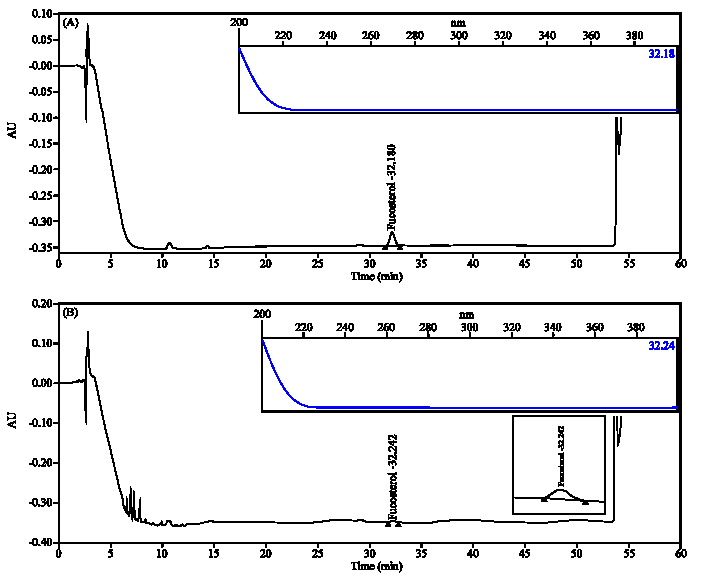
In vitro Cytotoxic Activity of Sargassum thunbergii and Dictyopteris divaricata (Jeju Seaweeds) on the HL-60 Tumour Cell Line
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
Y.M. Ham
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
J.Y. Moon
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
M.J. Kim
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
D.S. Kim
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
W.J. Lee
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea
N.H. Lee
Jeju Seaweed Industry Development Agency, Korea
C.G. Hyun
Jeju Biodiversity Research Institute (JBRI), Jeju High-Tech Development Institute (HiDI), Jeju 699-943, Korea