Research Article
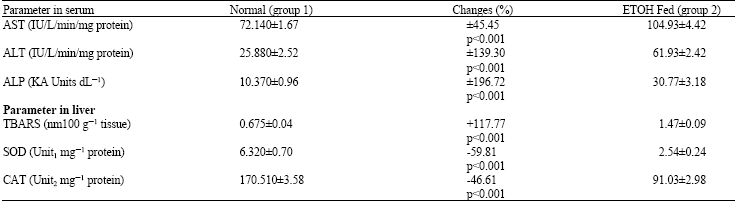
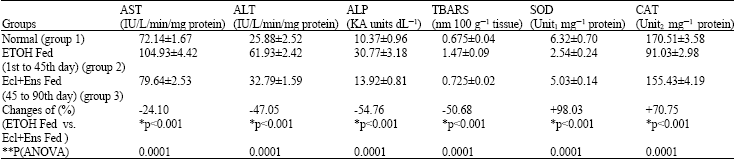
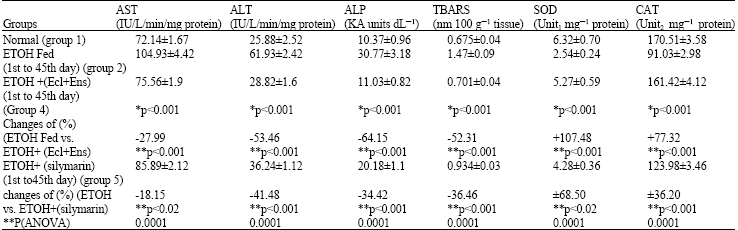
Hepatoprotective Effect of Enicostemma littorale blume and Eclipta alba During Ethanol Induced Oxidative Stress in Albino Rats
Department of Biomedical Engineering, Rajiv Gandhi College of Engineering and Technology, Pondicherry-607402, India
E.K. Elumalai
P.G. and Research Department of Zoology, Voorhees College, Vellore-632001, Tamilnadu, India
C. Sivakumar
P.G. and Research Department of Zoology, Voorhees College, Vellore-632001, Tamilnadu, India
S. Viviyan Therasa
P.G. and Research Department of Zoology, Voorhees College, Vellore-632001, Tamilnadu, India
E. David
P.G. and Research Department of Zoology, Voorhees College, Vellore-632001, Tamilnadu, India