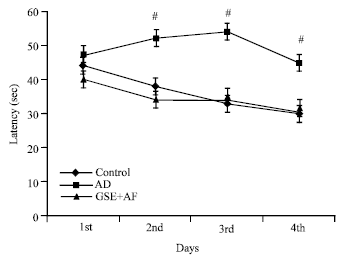
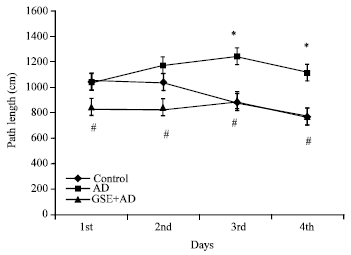
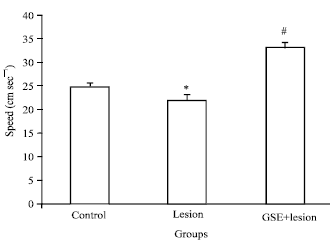
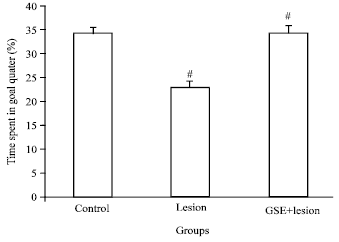
Research Article
Preventive Effect of Grape Seed Hydroalcholic Extract on Dementia Type of Alzheimer's Disease in Aged Male Rats
Physiology Research Center, Department of Physiology, Medical School, Ahwaz Jondishapour University of Medical Sciences, 61357-15794, Ahwaz, Iran
Sarkaki A.
Physiology Research Center, Department of Physiology, Medical School, Ahwaz Jondishapour University of Medical Sciences, 61357-15794, Ahwaz, Iran
Badavi M.
Physiology Research Center, Department of Physiology, Medical School, Ahwaz Jondishapour University of Medical Sciences, 61357-15794, Ahwaz, Iran