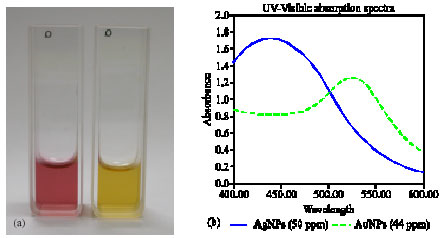
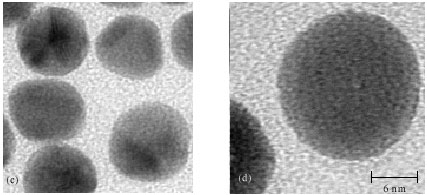
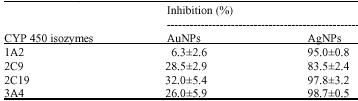
Research Article
Inhibition of Human Cytochrome P450 Enzymes by Metallic Nanoparticles: A Preliminary to Nanogenomics
Department of Anatomy, Faculty of Medicine, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand
P. Hongpiticharoen
Pharmaceutical Technology (International) Program, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand.
R. Rojanathanes
Department of Chemistry, Faculty of Science, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand
P. Maneewattanapinyo
Department of Chemistry, Faculty of Science, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand
S. Ekgasit
Department of Chemistry, Faculty of Science, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand
W. Warisnoicharoen
Pharmaceutical Technology (International) Program, Faculty of Pharmaceutical Sciences, Chulalongkorn University, Phyathai Road, Pathumwan, Bangkok 10330, Thailand.