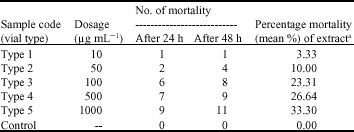
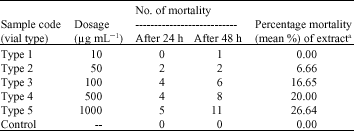
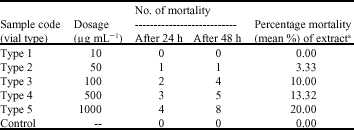
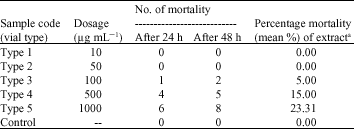
Research Article
Cytotoxicity Assessment of the Aerial Parts of Macrotyloma uniflorum Linn.
Department of Chemistry, Faculty of Science, University of Chittagong, Chittagong-4331, Bangladesh
E. Huq
Department of Chemistry, Faculty of Engineering, Bangladesh University of Engineering and Technology, Dhaka-1000, Bangladesh
N. Nahar
Department of Chemistry, Faculty of Science, University of Dhaka, Dhaka-1000, Bangladesh