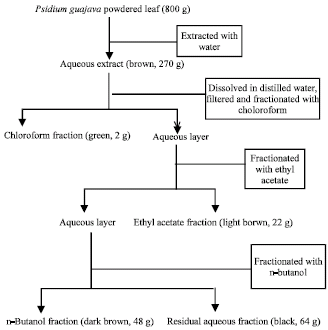
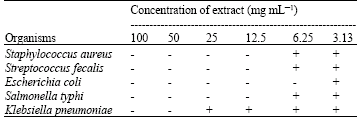
Research Article
Phytochemical Screening and Antibacterial Properties of Organic Solvent Fractions of Psidium guajava Aqueous Leaf Extracts
Department of Veterinary Medicine, University of Maiduguri, P.M.B 1 069: Maiduguri, Nigeria
A. G. Ambali
Department of Veterinary Medicine, University of Maiduguri, P.M.B 1 069: Maiduguri, Nigeria
P. A. Onyeyili
Department of Veterinary Physiology and Pharmacology, University of Maidugurl, P.M.B. 1069, Maiduguri, Nigeria