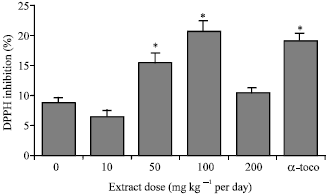
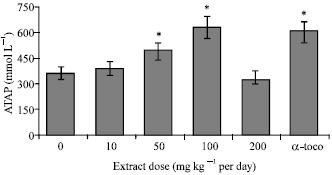
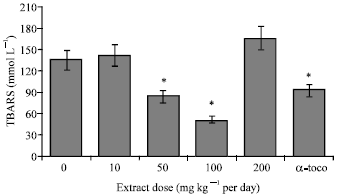
Research Article
On the Anti Oxidative Stress Potential of Zataria multiflora Boiss (Avishan shirazi) in Rats
Laboratory of Toxicology, Faculty of Pharmacy and Pharmaceutical Sciences Research Centre, Tehran University of Medical Sciences, Tehran, Iran
Narguess Yasa
Laboratory of Pharmacognosy, Faculty of Pharmacy and Medicinal Plants Research Center, Tehran University of Medical Sciences, Tehran, Iran
Azadeh Mohammadirad
Laboratory of Toxicology, Faculty of Pharmacy and Pharmaceutical Sciences Research Centre, Tehran University of Medical Sciences, Tehran, Iran
Reza Khorasani
Laboratory of Toxicology, Faculty of Pharmacy and Pharmaceutical Sciences Research Centre, Tehran University of Medical Sciences, Tehran, Iran
Mohammad Abdollahi
Laboratory of Toxicology, Faculty of Pharmacy and Pharmaceutical Sciences Research Centre, Tehran University of Medical Sciences, Tehran, Iran