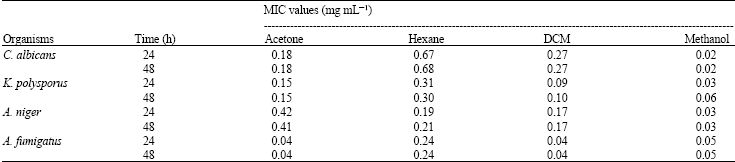
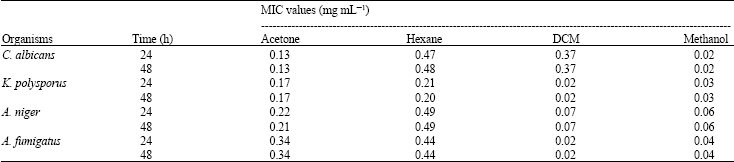
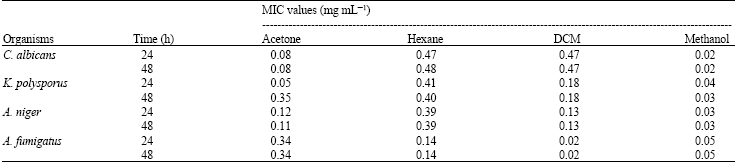
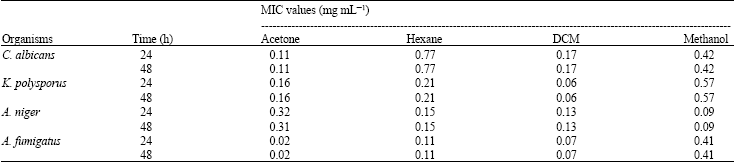
Research Article
Assessment of Antifungal Activity of Some Medicinal Plants
Division of Plant Biotechnology, Department of Botany, Karnatak University, Pavate Xagar, Dharwad-580003, Karnataka State, India
S. Vijay Kumar
Department of Biotechnology, Madanpalle Institute of Technology and Science, Madanpalle-517325, Chitoor District, Andhra Pradesh, India