Research Article
Effect of Calcium Chloride on Cyclophosphamide-Induced Genotoxic and Biochemical Changes in Swiss Albino Mice
Department of Pharmacology, College of Pharmacy king Saud University, P.O. Box 2457, Riyadh-11451 Saudi Arabia
Calcium chloride is indispensable in a variety of industrial and medicinal uses, in addition to its importance as a dietary supplement. It is reported to have low toxicity and is generally recognized as safe (GRAS) by the U.S. Food and Drug Administration (UNEP publications, 2002a). Calcium is useful in the formation of skeletons, neural transmission, muscle contraction and coagulation of the blood, in addition to its known treatment against hypocalcemic tetany, laryngospasm and osteoporosis (Marcus, 1996). The chloride ions are vital micronutrient in photosynthesis and osmoregulation of plants and are required for normal cellular functions in both animals and humans (UNEP publications, 2002b).
Calcium chloride is well known for its protective activity against some toxicants. Furihata et al. (1989) reported calcium chloride to inhibit stimulation of replicative DNA synthesis induced by sodium chloride in pyloric mucosa of male Fischer 344 rats. The mutagenic activity and cytotoxicity of dimethylnitrosamine (DMN) in CHO/HGPRT system (Chinese hamster ovary cell/Hypoxanthine-guanine phosphoribosyl transferase system) were greatly increased, upon addition of calcium chloride to rat liver metabolic activation system (S9) buffered with sodium phosphate (Li, 1984). Aboul-Ela (2002) found it to protect against the chromosomal aberrations and spermatozoa abnormalities caused by lead acetate.
Cyclophosphamide is an alkylating agent that is commonly used as an anticancer and an immunosuppressant drug. Although, CP is a known mutagenic and a pro-oxidant agent (Al-Bekairi et al., 1992; Selvakumar et al., 2006; Kumar et al., 2007; Zhang et al., 2007); It is often used for the treatment of preparation of bone marrow transplantation, B-cell chronic lymphocytic leukemia, myeloid leukemia, acute leukemias, lymphomas and breast cancers alone or in combination with other cytotoxic drugs (Ferry and Socie, 2003; Martin et al., 2003; Kowal et al., 2004). The mutagenic nature of cytotoxic drugs is known to cause secondary affliction and is a major drawback for the long term survivors of chemotherapy (Zounkova et al., 2007).
The present study on the protective effect of calcium chloride against the genotoxic and biochemical effects of CP was undertaken in view of the literature reports on the (i) immense use of calcium chloride as a food supplement, (ii) genotoxicity of anticancer agents and literature reports on protection of clastogencity by calcium chloride and (iii) a paucity of reports on the protection of genotoxicity of CP by calcium chloride.
The present study on the effect of calcium chloride on cyclophosphamide-induced genotoxic and biochemical changes in Swiss albino mice was conducted in the Department of Pharmacology, College of Pharmacy, King Saud University. The experimental part was undertaken during the period June to September 2007.
Chemicals: Calcium chloride (CAS No. 10043-52-4) obtained from Sigma Chemical Co., St. Louis, MO, USA., was used as the test compound. All the other chemicals and reagents were of analytical grade purchased from commercial sources. Cyclophosphamide purchased from ASTA Pharma A.G., (F.R.G.), was used as a standard drug.
Animals: Swiss albino male mice (SWR, home bred), aged 6-8 weeks and weighing 26-30 g, provided by Experimental Animal Care Center, College of Pharmacy, King Saud University were used in the study. All experimental mice were provided with Purina chow and free access to water. The animals were maintained under controlled conditions of temperature, humidity and light. The conduct of experiments and the procedure of sacrifice (using ether) were approved by the Ethics Committee of the Experimental Animal Care Society, College of Pharmacy, Riyadh, Kingdom of Saudi Arabia.
Dose selection, preparation of drugs, route and duration of administration: The dose of CP was based on earlier studies in our laboratory (Al-Bekairi et al., 1992; Qureshi et al., 1993). The doses of calcium chloride selected were 50, 100 and 200 mg/kg/day determined on the basis of (i) trial experiments conducted, which showed it to be pharmacologically effective at a dose of 100 mg kg-1(ii) reports in the literature (Wong et al., 1991; Aboul-Ela, 2002) and (iii) Human Therapeutic Dose (HTD) with reference to the surface area rule. The average intake of calcium chloride has been estimated to be 160-345 mg/kg/day for humans (UNEP publications, 2000a). According to the rule of surface area ratio of mice (20 g) and human (60), the calculated ratio is 0.0026 and hence the dose of calcium chloride (per 50 mice weighing 1 kg) for the lower limit of HTD would be (0.0026x160.0x50 = 20.80 mg/kg/day) and the upper limit would be (0.0026x345.0x50 = 44.85 mg/kg/day). The different doses (50-200 mg/kg/day) of calcium chloride used in the present study were more than the calculated doses. The increased doses used in the present experiment were in view of the reports that the metabolic rate in mice is six times (20.80x6 = 124.8) and 44.85x6 = 269.0 mg/kg/day) more than the calculated value as compared to human beings (Al-Yahya et al., 2006). Single dose of CP was given intraperitoneally (i.p.) in tap water (0.1 mL/10 g body weight of mice). Aqueous solution of calcium chloride was administered (24 h apart for 7 days) i.p. The animals in the control group were administered the vehicle by gavage (0.1 mL/10 g body weight of mice).
Experimental groups: A total of 40 male mice were used in different control and treatment groups of the micronucleus test (5 mice in each group) carried out in the study. Another 40 animals were used to conduct experiments on biochemical changes in proteins, nucleic acids (5 mice in each group) and 40 animals were used for the conduct of experiments on estimation of MDA and NP-SH (5 mice in each group). The experimental groups of mice consisted of: (T1) untreated control (tap water); (T2) calcium chloride 50.00 mg/kg/day; (T3) calcium chloride 100.00 mg/kg/day; (T4) calcium chloride 200.00 mg/kg/day; (T5) CP 100 mg kg-1 i.p.; (T6) calcium chloride 50.00 mg/kg/day, pretreatment (7 days) + CP 100 mg kg-1 i.p.; (T7) calcium chloride 100.00 mg/kg/day, pretreatment (7 days) + CP 100 mg kg-1 i.p.; (T8) calcium chloride 200.00 mg/kg/day, pretreatment (7 days) + CP 100 mg kg-1 i.p. Calcium chloride was administered orally to groups 2, 3, 4, 6, 7 and 8 for 7 days. CP was injected 24 h before sacrifice. In each case animals were killed 24 h after the last treatment, femurs excised for cytological studies by micronucleus test. Livers were excised and preserved at -70°C for the analysis of biochemical changes.
Micronucleus test: The procedure described by Schmid (1975) was used. From the excised femurs, cells were collected in foetal calf serum. After centrifugation the cells were spread on slides and air-dried. Coded slides were fixed in methanol and stained with May-Gruenwald solution followed by Giemsa staining. The polychromatic erythrocytes (PCE) were screened for micronuclei and the normochromatic erythrocytes (NCE) in order to obtain a PCE/NCE ratio to analyze the mitodepression (bone marrow depression).
Estimation of total proteins: Total proteins were estimated by the modified Lowry method of Schacterle and Pollack (1973). Bovine serum albumin was used as standard.
Determination of nucleic acids: The method described by Bregman (1983) was used to determine the levels of nucleic acids. Tissues were homogenized and the homogenate was suspended in ice-cold trichloroacetic acid (TCA). After centrifugation, the pellet was extracted with ethanol. DNA was determined by treating the nucleic acid extract with diphenylamine reagent and reading the intensity of blue color spectrophotometrically at 600 nm. For quantification of RNA, the nucleic acid extract was treated with orcinol and the green colour was read spectrophotometrically at 660 nm. Standard curves were used to determine the amounts of nucleic acids present.
Malondialdehyde estimation: The method described by Ohkawa et al. (1979) was used. Malondialdehyde (MDA) was measured as an indicator of lipid peroxidation. Cardiac and liver tissues were homogenized in potassium chloride solution and incubated with thiobarbituric acid. After centrifugation the pink clear layer was read spectrophotometrically at 532 nm. Malondialdehyde bis (dimethyl acetal) was used as an external standard.
Determination of nonprotein sulfhydryl groups (NP-SH): The estimation of NP-SH levels in different organs was undertaken according to the method of Sedlak and Lindsay (1968). The tissues were homogenized in ice cold 0.02 M ethyleneodiaminetetraacetic acid disodium. The homogenate was treated with 50% w/v trichloroacetic acid and centrifuged. Supernatant fractions were mixed with tris buffer, 5-5’-dithiobis-(2 nitrobenzoid acid) (DTNB) was added. After shaking the contents, its absorbance was determined spectrophotometrically at 412 nm within 5 min of the addition of DTNB against reagent blank with no homogenate.
Statistical analysis: Data are presented as mean±SE. Statistical comparison were made by using analysis of variance (ANOVA) with significance defined as p<0.05. Post-hoc analyses were done by using Student’s t-test.
The results showing the effect of calcium chloride on micronucleated polychromatic erythrocytes (M-PCE) and the ratio of polychromatic to normochromatic cells (PCE/NCE) indicating mitodepression after 24 h of treatment are presented in Table 1. The frequency of M-PCE observed in untreated mice (group 1) and treatment with calcium chloride at 50, 100 and 200 mg/kg/day (groups 2, 3 and 4) for 7 days were found to be in the same range. The PCE/NCE ratio in groups 2, 3 and 4 was also in the same range except the high dose (group 4) of calcium chloride where the values were slightly less at the high dose; however, the difference was negligible when compared to mitodepression induced by CP. CP treatment (group 5) was found to induce a significant increase in the frequency of micronuclei (p<0.001). The ratio of PCE/NCE was suppressed (p<0.001) after treatment with CP as compared to controls.
The pretreatment with calcium chloride at low dose (group 6) failed to prevent the CP-induced frequency of micronuclei and bone marrow depression as compared to CP treatment (group 5). The frequency of micronuclei induced by CP was inhibited in mice pretreated with both the medium (group 7, p<0.05) and high doses (group 8, p<0.01) of calcium chloride. The PCE/NCE ratio was not significantly altered in any of the pretreatment groups (6, 7 and 8) when compared to mitodepression induced by CP (group 5) (Table 1).
Treatment with calcium chloride for 7 days failed to show any significant changes in the concentrations of proteins, RNA and DNA, while CP treatment significantly decreased the hepatic levels of proteins (p<0.01), RNA and DNA (p<0.05). The pretreatment with calcium chloride for 7 days failed to protect the CP-induced depletion of proteins and RNA, whereas, the DNA concentrations were significantly (p<0.05) protected at the high dose (200 mg/kg/day) of calcium chloride against the treatment with CP (Table 2).
| Table 1: | Effect of calcium chloride on cyclophosphamide-induced inhibition in the frequency of micronuclei and the ratio of femoral erythrocytes in male Swiss albino mice |
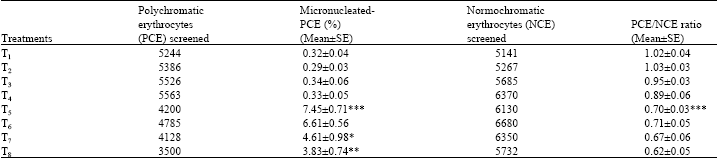 | |
| Fifteen mice were used in each group; *: p<0.05; **: p<0.01; ***: p<0.001 (One way ANOVA and Post hoc Tukey-Kramermultiple comparison test was done) | |
| Table 2: | Effect of calcium chloride on cyclophosphamide-induced changes in Protein and Nucleic acid concentrations of hepatic tissue in Swiss albino mice. |
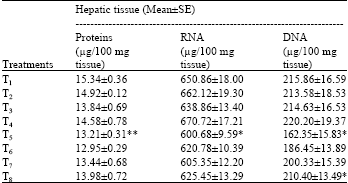 | |
| Five mice were used in each group; Groups 2, 3 and 4 were statistically compared with group 1 and groups 6, 7 and 8 were statistically compared with group 5; *p<0.05; **p<0.01 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done) | |
The treatment with calcium chloride for 7 days (groups 2-4) failed to induce any significant changes in the hepatic levels of MDA as compared to the values obtained in the control (group 1). CP treatment (group 5) increased the MDA levels significantly (p<0.001) as compared to group 1. The pretreatment with calcium chloride for 7 days was found to significantly reduce the levels of MDA (p<0.05, group 8) as compared to the value obtained in the CP group (Table 3).
The hepatic levels of NP-SH were not affected by the treatment for 7 days with the different doses of calcium chloride (groups 2-4), whereas the treatment with CP (group 5) significantly (p<0.001) decreased these concentrations as compared to the values obtained in the control (group 1). Pretreatment with calcium chloride for 7 days was found to significantly (p<0.05) increase the NP-SH concentrations at the high dose (group 8) as compared to the values obtained after treatment with CP (Table 3).
The results obtained in the present study showed that the treatment with calcium chloride did not induce micronuclei and has little or no bone marrow suppression after 7 days of treatment. These results clearly indicate lack of any clastogenic and/or cytotoxic activity caused by calcium chloride. CP treatment was found to be highly clastogenic and showed significant cytotoxicity. The known clastogenic and cytotoxic potentials of CP (Qureshi et al., 1993; Selvakumar et al., 2006; Kumar et al., 2007) were thus confirmed by this study. The observed clastogenicity of CP may be attributed to its prooxidant properties (Zhang et al., 2007). It is well known that various anticancer drugs have clastogenic activities and induce micronuclei formation (Matlawska-Wasowska et al., 2007; Zounkova et al., 2007).
| Table 3: | Effect of calcium chloride on cyclophosphamide-induced changes in MDA and NP-SH concentrations of hepatic tissue in Swiss albino mice |
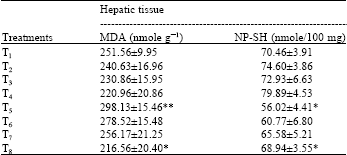 | |
| Five mice were used in each group; Groups 2, 3 and 4 were statistically compared with group 1 and groups 6, 7 and 8 were statistically compared with group 5; *p<0.05; **p<0.01 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done) | |
Micronuclei are derived from chromosomal fragments or chromosomes which are not included in the daughter nuclei at cell division and their frequency depends on the rate of chromosomal breakage or of chromosomal loss (Roy et al., 2005). The exact mode of formation of micronuclei is not known, however; there is experimental evidence to show that induction of clastogenicity and/or cytotoxicity might be related to the formation of adducts or breaks of DNA (Cullinane et al., 2000). The induction of micronuclei and the cytotoxicity caused by CP were found to be inhibited by the pretreatment of calcium chloride. These results support the observation of Aboul-Ela (2002) who found calcium chloride to protect against the chromosomal aberrations caused by lead acetate. However, present results contradicts the results of Li (1984), who found the mutagenic activity and cytotoxicity of dimethylnitrosamine (DMN) in CHO/HGPRT system (Chinese hamster ovary cell/Hypoxanthine-guanine phosphoribosyl transferase system) were greatly increased, upon addition of calcium chloride to rat liver metabolic activation system (S9) buffered with sodium phosphate (LI, 1984). The discrepancy between the results might be due to the in vitro nature of study conducted by Li (1984). The different results obtained in our study may be due to the exposure of the drug to pharmacokinetic, pharmacodynamic and cell cycle processes involved in the whole organ (in vivo), which is different from the artificial conditions that occur in cells in vitro (Aaron et al., 2002; Lullmaun et al., 1993). Furthermore, the procedures used in the in vitro studies have their own shortcomings. The processes of homogenization, cell disruption and membrane vesicle fragmentation may result in the rearrangement and alteration of P450 systems which are known to produce reactive oxygen species (Halliwell and Gutteridfge, 1987b; Koop, 2006).
Decreases in the ratio of PCE to mature erythrocytes are considered an indication of mutagen induced bone marrow cytotoxicity (Al-Majed et al., 2006). While there was no bone marrow depression observed after treatment of calcium chloride at the doses used in the present study, CP treatment was found to decrease the ratio of PCE to mature erythrocytes. CP treatment also inhibited the contents of nucleic acids and proteins in the hepatic cells, which might have contributed to the observed cytotoxicity of CP, besides production of free radicals (Zhang et al., 2007). Pretreatment with calcium chloride was found to protect against induction of micronuclei, bone marrow depression and depletion of DNA caused by CP. Our results are in agreement with the observation of Furihata and Matsushima (1990) who found calcium chloride to inhibit sodium chloride-induced replication of DNA synthesis in pyloric mucosa of F344 rats. Although, there is no direct literature on cytotoxic activity of calcium chloride, it has been shown to protect against the carcinogenicity and toxicity of other compounds. Nishikawa et al. (1992) found calcium chloride to inhibit the post-initiation phase of two-stage glandular stomach carcinogenesis in rats induced by N-methyl-N’-nitro-N-nitrosoguanidine (MNNG) and sodium chloride.
Antineoplastic drugs in general are known to increase the load of free radicals and stimulate lipid peroxidation (MDA) in the body (Halliwell and Gutteridfge, 1987a). MDA is a compound that increases oncogenic potentials (Borek, 1988; Surapaneni and Venkata, 2006) and hence any rise in the levels of MDA is deemed to be associated with cancer. Our result on increase in the hepatic levels of MDA caused by CP support an earlier report (Berrigan et al., 1987) which showed it to increase lipid peroxidation in hepatic cell membrane. Al-Nasser (1998) also found CP to cause hepatotoxicity. In the present study, the pretreatment with calcium chloride was found to decrease the CP-induced increase in the concentrations of MDA. This reduction in the levels of MDA might be related to the antioxidative potentials of calcium chloride. These results confirm observation of a previous report, which showed calcium chloride to reduce the MNNG/sodium chloride-induced increase in the levels of MDA in gastric mucosa of rats (Nishikawa et al., 1992). Glutathione (NP-SH) is a known scavenger of hydroxyl radicals and singlet oxygen. In the present investigation, the level of NP-SH in hepatic cells was significantly reduced after treatment with CP. The pretreatment with calcium chloride was found to protect the inhibition of NP-SH caused by CP. These results support the antioxidant potentials of calcium chloride (Shin et al., 2006; Sheweita and Khoshhal, 2007).
Taken together, present results demonstrate that pretreatment with calcium chloride (i) protected against the CP-induced increase in the frequency of micronuclei and depletion of DNA without any alteration in its cytotoxicity and (ii) inhibited the CP-related increase and depletion of lipid peroxidation and glutathione, respectively. These data show that calcium chloride might be useful to avert secondary tumor risk by decreasing the accumulation of free radicals.