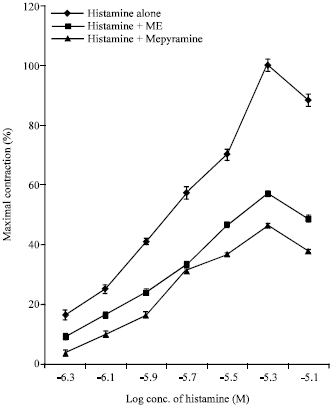
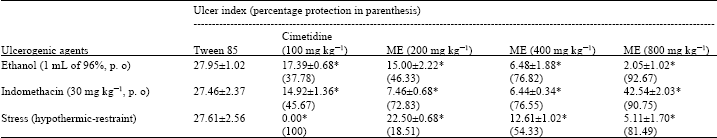
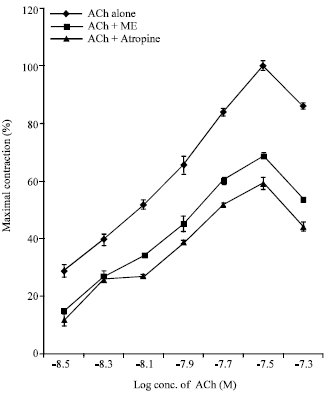
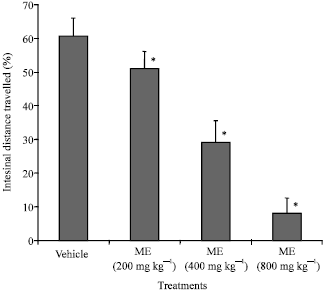
Research Article
Gastro-Protective Properties of the Leaf Extracts of Ocimum gratissimum L. Against Experimental Ulcers in Rat
Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, 410001, Enugu State, Nigeria
Lucy John- Africa
Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, 410001, Enugu State, Nigeria
C. S. Nworu
Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, University of Nigeria, Nsukka, 410001, Enugu State, Nigeria