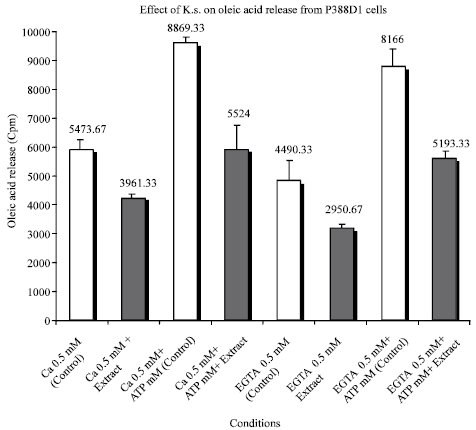
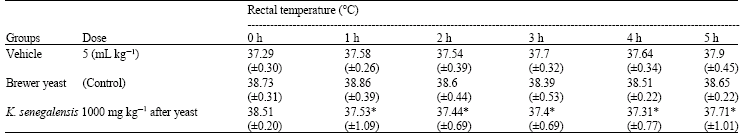
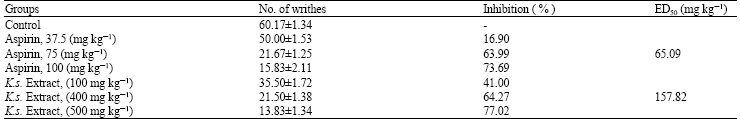
Research Article
Mechanism of the Anti-inflammatory Activity of Khaya senegalensis A. Juss. (Meliaceae)
Pharmacology and Toxicology Laboratory of the Health Sciences Research Institute (RSS/CNRST), 03 BP 7192 OUAGADOUGOU 03, Burkina, Faso
I. P. Guissou
Pharmacology and Toxicology Laboratory of the UFR/SDS University of Ouagadougou 03, BP 7021 OUAGADOUGOU Burkina, Faso
J. Dubois
Laboratory of Bioanalytical Chemistry, Toxicology and Applied Physical Chemistry, CP 205/1 - Institute of Pharmacy, Free University of Brussels
J. P. Dehaye
General and Human Biochemistry Laboratory, CP 205/3 Institute of Pharmacy, Free University of Brussels, Belgium
S. Ouedraogo
Pharmacology and Toxicology Laboratory of the Health Sciences Research Institute (RSS/CNRST), 03 BP 7192 OUAGADOUGOU 03, Burkina, Faso
A. Traore
Pharmacology and Toxicology Laboratory of the Health Sciences Research Institute (RSS/CNRST), 03 BP 7192 OUAGADOUGOU 03, Burkina, Faso
N. Some
Pharmacology and Toxicology Laboratory of the Health Sciences Research Institute (RSS/CNRST), 03 BP 7192 OUAGADOUGOU 03, Burkina, Faso