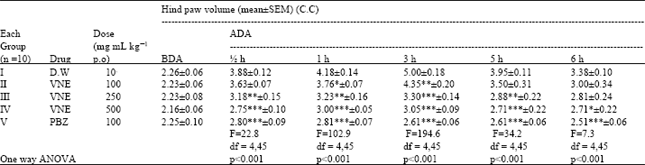
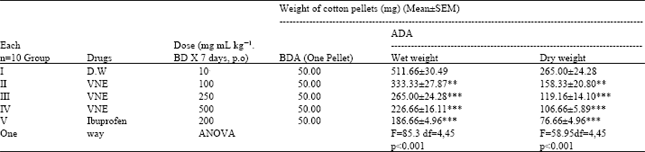
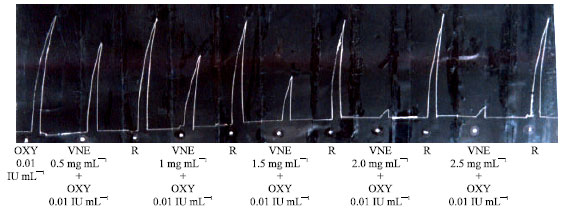
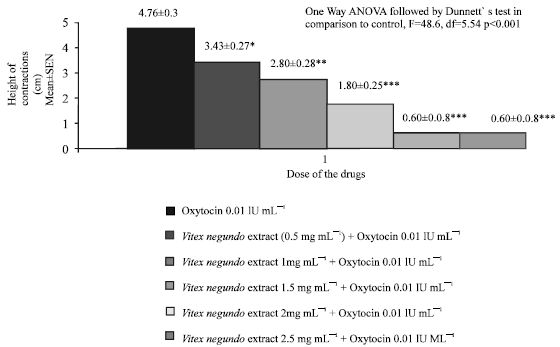
Research Article
Anti-inflammatory Activity and Mechanism of Action of Vitex negundo Linn
Post Graduate Department of Pharmacology and Therapeutics, Government Medical College Jammu (J and K) India-1
Rajesh Kumar Gupta
Department of Pharmacology, Mahatma Gandhi Institute of Medical Sciences (Sevagram), Wardha, Maharashtra, India