Research Article
Genetic and Biochemical Toxicity of Guarana After Sub-Acute Treatment in Somatic and Germ Cells of Swiss Albino Mice
Department of Pharmacology, College of Pharmacy king Saud University, P.O. Box 2457, Riyadh-11451 Saudi Arabia
Paulinia cupana (Guarana) is a Brazilian plant that belongs to the family ‘Sapindacea’. The plant has folkloric significance because of its caffeine contents and stimulant property. It is useful for the production of stimulants, soft drinks and sweets. In the drug scene it is sometimes trafficked as a natural stimulant or drug surrogate (Schafer, 1999). Dietary supplements that contain Guarana are widely marketed and used in the USA for weight loss and athletic performance enhancement. Studies on weight loss showed herbal drug (Yerbe Mate) containing Guarana seeds and Damiana leaves, induced significant weight loss over 45 days in overweight patients (Andersen and Fogh, 2001). Boozer et al. (2001) found herbal supplement containing Ma Huang, Guarana and other ingredients are effective in weight loss in human subjects.
A large number of reports are available on the clinical toxicity of some compound preparations containing Guarana and/or Guarana alone. Mansi and Huang (2004) found medicines including Guarana, taken to reduce weight cause rhabdomyolysis. Serious cardiovascular toxicity is reported in people taking dietary supplements that contain ma huang and Guarana (Baghkhani and Jafari, 2002; Haller et al., 2002). Cannon et al. (2001) found Guarana to cause intractable ventricular fibrillation in humans. Studies on genotoxicity and mutagenicity showed extracts of Guarana to be genotoxic as assessed by lysogenic induction in Escherichia coli and they were also able to induce mutagenesis in Salmonella typhimurium (Fonseca et al., 1994). Nevertheless, these systems lack metabolizing enzymes involved in the metabolism of genotoxins (Dauer et al., 2003). The present study was undertaken in view of (I) the limited predictive value of procaryotic test systems and (ii) a paucity of literature on genotoxicity and biochemical toxicity of Guarana in somatic and germ cells in rodents.
The present study on genetic and biochemical effects of Guarana was conducted in the Department of Pharmacology, College of Pharmacy, King Saud University. The experimental part was undertaken during the period July 05 to October 05.
Test herbal product: Guarana was used as the test herbal product in the present study. It is manufactured by Natural Balance, Inc., Castle Rock, CO, 80104, USA and marketed in form of capsules by General Nutrition Corporation (GNC) of USA in Saudi Arabia. Each capsule contains proprietary blend weighing 531 mg. The blend consists of Guarana (standardized seed extract yielding 72 mg of caffeine) ephedra (standardized plant body extract yielding 12 mg of ephedrine), passion flower (aerial portion extract), gotu kola (aerial portion extract), wood betoni (aerial portion). The other ingredients are magnesium stearate and gelatin.
Dose selection and route of administration: The doses selected for conduct of different studies were based on the LD50 (2.12 g kg-1) value (evaluated in our laboratory) and preliminary experiments. The different doses selected for Guarana were 133, 265 and 530 mg kg-1, body weight/day, corresponding 1/16, 1/8 and 1/4, respectively of the LD50 (Wallace Hayes, 1989). The duration of treatment was 7 days (sub-acute). The dosage form was aqueous suspension and the route of administration, gastric intubation (oral) in all the experiments.
Animal stocks: Male Swiss albino mice (SWR) aged 6-8 weeks and weighing 25-28 g were obtained from the Experimental Animal Care Center, King Saud University, Riyadh, Saudi Arabia. The animals were fed on Purina chow diet and water ad libitum and were maintained under standard conditions of humidity, temperature and light (12 h, light/12 dark cycle). The conduct of experiments and the procedure of sacrifice (using ether) were approved by the Ethics Committee of the Experimental Animal Care Society, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia.
Experimental groups: The experimental groups of mice consisted of the following: group 1, control (0.3 mL/mouse, tap water); Group 2, Guarana (133 mg/kg/day); Group 3, Guarana 265 mg/kg/day); group 4, Guarana (530 mg/kg/day). A total of 100 mice were used in the experiment as follows: (I) Cytological studies on micronucleus test (20 mice), (ii) Evaluation of spermatozoa abnormalities (20 mice), (iii) Evaluation of the chromosomal aberrations in the testes (20 mice), (iv) biochemical study on estimation of proteins, nucleic acids, MDA and NP-SH) in hepatic cells (20 mice) and (v) biochemical study on quantification of proteins, nucleic acids, MDA and NP-SH in testicular cells (20 mice).
Cytological studies on micronucleus test: The procedure of micronucleus test described by Schmid (1975) was followed. The mice were sacrificed 24 h following 7 day treatment with Guarana. The femoral cells were collected in fetal calf serum. After centrifugation, the cells were spread on slides and air-dried. Coded slides were fixed in methanol and stained in May-Gruenwald solution followed by Giemsa stain. The polychromatic erythrocytes (PCE) were screened for micronuclei, and reduction of the mitotic index was assessed on the basis of the ratio of polychromatic erythrocytes/normochromatic erythrocytes (PCE/NCE).
Evaluation of spermatozoa abnormalities: The mice were sacrificed 5 weeks after the last day of the seven day treatment (Wyrobek and Bruce, 1975; Al-Shabanah, 1997). The different spermatozoa abnormalities (amorphous, banana shaped, swollen achrosome, triangular head, macrocephali and rotated head) screened were those found in all the s lides (Wyrobek et al., 1983; Anderson et al., 1983).
Evaluation of the chromosomal aberrations in the testes: In analysis of the chromosomal aberrations, the mice were sacrificed 19 days after the last day of the treatment (Schleismacher, 1970; Qureshi et al., 1988; Al-Shabanah, 1997). The chromosomal preparations were made by the air drying technique (Evans, 1964; Al-Shabanah, 1997). The coded slides were stained in Giemsa solution and screened for the aberrations including aneuploids, autosomal univalents, sex-univalents and polyploids.
Biochemical evaluation: The mice were sacrificed 24 h following 7 day treatment with Guarana. The liver and testes were quickly excised and freezed at -70°C. The frozen samples were used for estimation of proteins, RNA, DNA, MDA and NP-SH levels.
Estimation of total proteins and nucleic acids: Total proteins were estimated by the modified Lowry method of Schacterle and Pollack (1973). Bovine serum albumin was used as a standard. The method described by Bregman (1983) was used to determine the levels of nucleic acids.
Determination of MDA concentrations: The method described by Ohkawa et al. (1979) was used. The tissue was homogenized in TCA solution and the homogenate suspended in thiobarbituric acid. After centrifugation the optical density of the clear pink supernatant was read at 532 nm. Malondialdehyde bis (dimethyl acetal) was used as an external standard.
Quantification of the NP-SH levels: The method described by Sedlak and Lindsay (1968) was used to determine the levels of NP-SH. The tissue was homogenized in ice cold 0.02 M ethylene-o-amine tetra acetic acid disodium (EDTA) and mixed with TCA. The homogenate was centrifuged at 3000 g. The supernatant was suspended in tris buffer, 5-5’-dithiobis-(2 nitrobenzoic acid) (DTNB) and read at 412 nm against reagent blank with no homogenate.
Statistical analysis: One way ANOVA and Post hoc Tukey-Kramer multiple comparison tests statistically analyzed the different studies undertaken.
The sub-acute treatment with Guarana caused a dose-dependent increase in the frequency of micronucleated-PCE, however; the increase was statistically significant (p<0.05) at the higher dose (530 mg/kg, body weight/day). The treatment failed to cause any changes in the PCE/NCE ratio (p>0.05) as compared to the values in the control group (Fig. 1). The ratio was found to be higher at the lower doses, while at the high dose (530 mg/kg body weight/day) it reduced. There was an increase observed in the proportion of sperm abnormalities such as swollen achrosome, macrocephali and total abnormalities, however; these changes were statistically insignificant as compared to the values observed in the control group (Fig. 2). A significant (p<0.05) increase was observed in the frequency of aneuploids and total percent aberrations (265 and 530 mg/kg body weight/day) and sex-univalents (530 mg/kg, body weight/day) as compared to the values observed in the control (Fig. 3).
The results of biochemistry showed that sub-acute treatment with Guarana caused a dose dependent decrease in the hepatic levels of RNA and DNA. The depletion was statistically significant (p<0.05) in these concentrations at the higher dose (530 mg/kg body weight/day) as compared to the values observed in the control (Table 1). The testicular levels of proteins, RNA and DNA were decreased at the high dose (530 mg/kg body weight/day) of Guarana as compared to the values observed in the control. The changes in these levels observed at the lower doses were not prominent (Table 1). The hepatic levels of MDA and NP-SH were significantly increased (p<0.01) and decreased (p<0.05), (respectively) after the sub-acute treatment with Guarana at the higher dose (530 mg/kg body weight/day) (Table 2), while at the lower doses, these changes were insignificant. There was a dose-dependent and significant increase in the testicular concentrations of MDA concentrations after treatment with Guarana (p<0.05) at 133, (p<0.01) at 265 and (p<0.001) at 530 mg/kg body weight/day.
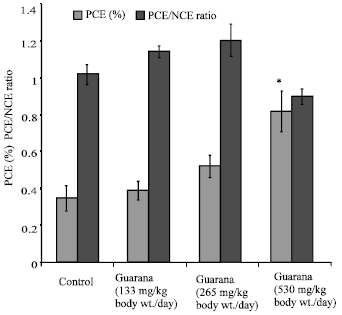 | |
| Fig. 1: | Effect of Guarana on the frequency of micronuclei and the ratio of polychromatic to normochromatic erythrocytes in femoral cells of Swiss albino mice after sub-acute treatment. Five mice were used in each group, *p<0.01 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done individually for different paramaters) |
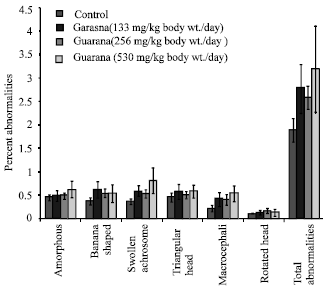 | |
| Fig. 2: | Effect of Guarana on epididymal spermatozoa in Swiss albino mice after sub-acute treatment. Five mice were used in each group p>0.05 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done individually for different paramaters) |
The testicular levels of NP-SH were significantly (p<0.05) decreased at the higher dose (530 mg/kg body weight/day) only, as compared to the values obtained in the control group (Table 2).
| Table 1: | Effect of Guarana on protein and nucleic acid contents in hepatic and testicular tissue of Swiss albino mice after Sub-acute treatment |
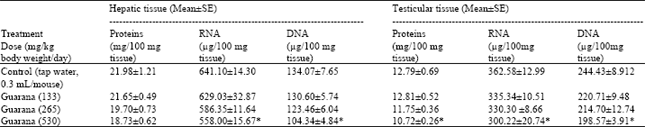 | |
| Five mice were used in each group. *p<0.05 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done individually for different parameters) | |
| Table 2: | Effect of Guarana on Malondialdehyde and Glutathione (NP-SH) concentrations in liver and testes of mice after sub-acute treatment |
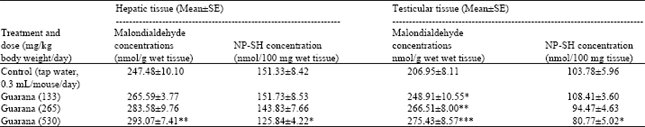 | |
| Five mice were used in each group. *p<0.05; **p<0.01; ***p<0.001. (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done individually for male, female and different parameters) | |
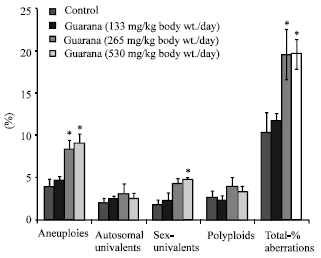 | |
| Fig. 3: | Effect of Guarana on testis chromosomes in Swiss albino mice after sub-acute treatment. Five mice were used in each group *p<0.01 (One way ANOVA and Post hoc Tukey-Kramer multiple comparison test was done individually for different paramaters) |
The treatment with Guarana significantly increased the frequency of micronucleated-PCE. These ata showed genotoxic effect of Guarana in femoral erythrocytes. The formation of micronuclei might be due to either clastogenic and/or aneugenic mechanisms. Our data on the frequency of micronucleated-PCE, corroborate with the results of Fonseca et al. (1994) that showed the genotoxic and mutagenic effects of Guarana in Escherichia coli and Salmonella typhimurium. In most of the studies, the Salmonella assay or other bacterial test systems were used for identifying the genotoxic activity of different compounds. However, the predictive value of procaryotic test systems is limited. Furthermore, these systems lack metabolizing enzymes involved in the metabolism of genotoxins and the addition of exogeneous metabolizing systems is necessary when promutagens are used (Dauer et al., 2003). Present results on micronucleus test is the first study in vivo on the genotoxicity of Guarana. These results are confirmed by present data on depletion of nucleic acids observed in the hepatic cells. The changes demonstrate clastogenic and cytotoxic potentials of Guarana. Present data is supported by the observation of Santa Maria et al. (1998) which demonstrated critical importance of guarana in dose-related cytotoxicity in in vitro bioassays. The clastogenic and cytotoxic potentials of Guarana might be related with the oxidant status in the liver cells, as revealed by variations in the hepatic concentrations of MDA and NP-SH. These results clearly indicate a weakening or failure of the antioxidant defense system in the hepatic cells. The lack of an antioxidant defense might have favored a heightened state of oxidative stress, thereby causing destruction of membranes and DNA (Kawamura et al., 1992; Scoltock et al., 2000) and induction of genotoxicity (Sapone et al., 2003; Blazovics, 2004; Ramprasath et al., 2005). Nevertheless, our study warrants the conduct of further experiments on molecular mechanisms.
Guarana treatment caused an increase in frequency of chromosomal abnormalities (aneuploids and sex-univalents) in the testes. The total chromosomal aberrations were also significantly increased. There is no parallel study relating the cytological effects on the germ cells. However, these results are in corroboration with our data on the depletion of proteins and nucleic acids in the testes. The depletion of nucleic acids is attributed to their oxidation, which obviously might have been resulted by the increase and decrease of MDA and NP-SH, respectively in the testicular cells, observed in our present study. These findings support previous reports, which suggested that the deficiency of endogenous antioxidants interfere with the meiosis and cause chromosomal anomalies (Wyrobek and Bruce, 1978; Watanabe and Endo, 1991; Al-Shabanah, 1997). However, these results contradict our data on the sperm abnormality, which showed lack of any effect of Guarana on morphology of the sperms. Although, there are no parallel studies available for a possible comparison, the data on testicular chromosomal aberrations, nucleic acids, MDA and NP-SH failed to reflect our observation on spermatozoa morphology. The discordance between the results on nucleic acids and the lack of spermatozoa abnormality might be related to the role of the excurrent duct system in eliminating morphologically abnormal spermatozoa during transit from testes to vas deferens and caudae epididymis. Previous studies (Perez-Sanchez et al., 1997; Chenoweth et al., 2000) have shown that the transit of spermatozoa through the extragonadal passage produces and/or eliminate morphologically abnormal spermatozoa by the excurrent duct system. The mechanism of these changes is yet a dilemma.
The genotoxic (somatic and germ cells), observed in the present study appears to be mediated through the reactive oxygen species. Our results are supported by recent studies, which have drawn a clear relation between the genesis of free radicals and the induction of genotoxicity (Sapone et al., 2003; Blazovics, 2004; Ramprasath et al., 2005). The constituent/(s) responsible for observed genetic changes are not known. Nevertheless, the observed genotoxicity may be related to tannin content of Guarana. Tannins are increasingly recognized as dietary carcinogens and as antinutrients interfering with the biological system’s full use of protein (Morton, 1992). They are also described as the DNA-damaging compounds (Dauer et al., 2003). Szakmary and Knasmuller (1991) found tannic acid to significantly increase the mosaic spots in Drosophila melanogaster (SMART-TEST). Tannic acid is also reported to cause an increase of micronuclei up to 50% in Hep G2 cells (Sanyal et al., 1997). Furthermore, condensed tannins have been described to cause induction of micronuclei in cultured V79 Chinese hamster cells (Grimmer et al., 1992). The exact mechanism of Guarana-induced genotoxicity is not known. However, besides the role of tannins, it might also be due to caffeine-related stimulation of sympathetic nervous system (Hoffman, 2001; Acheson et al., 2004) and enhancement of catcholamines and norepinephrine (Berlan et al., 1991; Poceta et al., 1994; Gyllenhaal et al., 1999; Swann et al., 2005) which are classified as oxidative mutagens (Martinez et al., 2000). These events have been associated with accumulation of reactive oxygen species, cytotoxicity and apoptosis (Hodach et al., 1975; Nishikawa et al., 1985; Eppler and Dawson, 2002; Khorchid et al., 2002; Fu et al., 2004) and impairement of DNA synthesis repair and integrity (Gilbert et al., 1977; Yokoyama et al., 1983; Kizaki et al., 1993; Fu et al., 2004).
In conclusion, the results of our present study clearly indicate genotoxic and biochemical toxicity of Guarana in both somatic and germ cells of mice. These changes might have serious implications that may (I) cause cellular proliferation and malignancy (ii) induce sterility, teratogenesis and dominant lethal mutations and (iii) increase the frequency of mutations in the exposed populations. Finally, it is suggested that, in view of the observed cytological and biochemical effects of Guarana, it may be subjected to a thorough evaluation of toxicity before it is available for human use.
The author is thankful to King Abdulaziz City for Science and Technology, Riyadh, Kingdom of Saudi Arabia, for sanction of grant to conduct research on herbal drugs (PROJECT AR-21-41).