Research Article
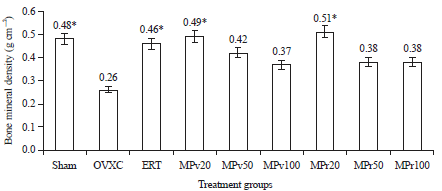
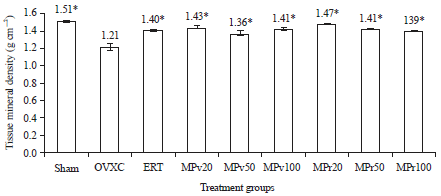
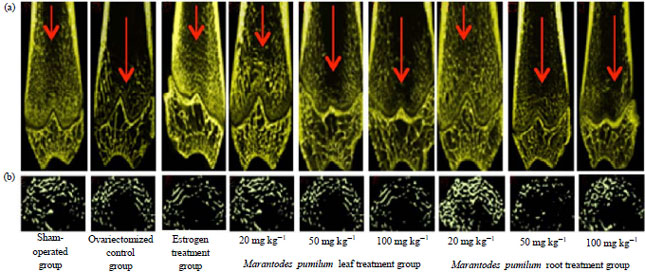
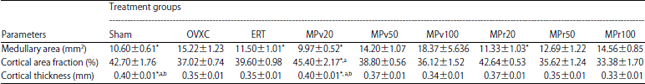
Low Dose Marantodes pumilum Leaf and Roots Extracts Preserved Bone Structure in Ovariectomized Rats
Department of Pharmacology, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
Ahmad Nazrun Shuid
Department of Pharmacology, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
LiveDNA: 60.1593
Ima Nirwana Soelaiman
Department of Pharmacology, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
Norliza Muhammad
Department of Pharmacology, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
Norazlina Mohamed
Department of Pharmacology, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
LiveDNA: 60.16153