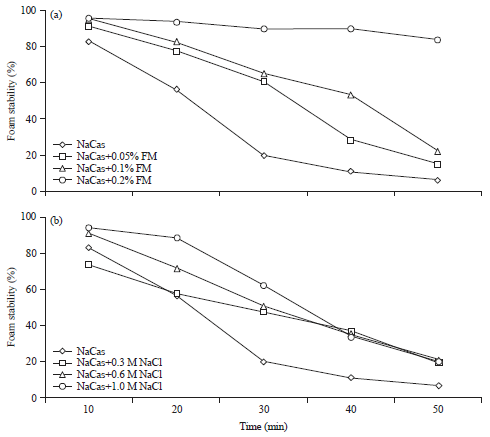
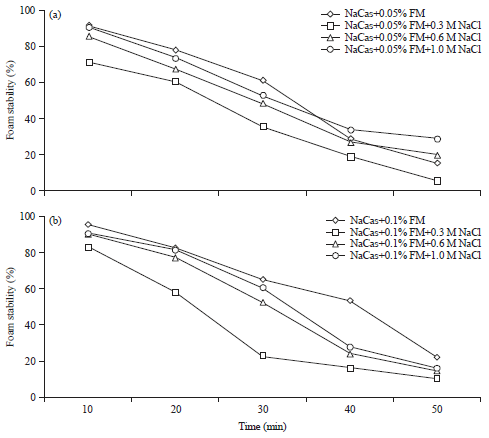
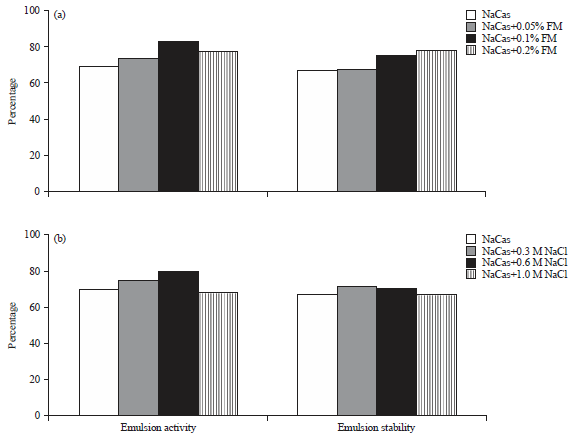
Research Article
Effect of Flaxseed Mucilage, Sodium Chloride and Their Combination on Some Functional Properties of Sodium Caseinate Solution
Dairy Department, Food Industries and Nutrition Research Division, National Research Centre, El-Buhouth St., Dokki, Cairo, Egypt
Ahmed B. Shazly
Dairy Department, Food Industries and Nutrition Research Division, National Research Centre, El-Buhouth St., Dokki, Cairo, Egypt
Mahmoud Abd El-Aziz
Dairy Department, Food Industries and Nutrition Research Division, National Research Centre, El-Buhouth St., Dokki, Cairo, Egypt
Tarek N. Soliman
Dairy Department, Food Industries and Nutrition Research Division, National Research Centre, El-Buhouth St., Dokki, Cairo, Egypt
LiveDNA: 20.23162