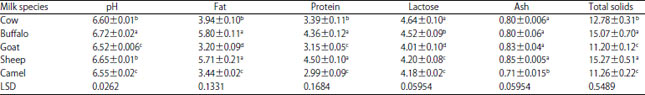
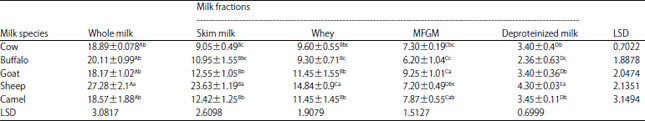
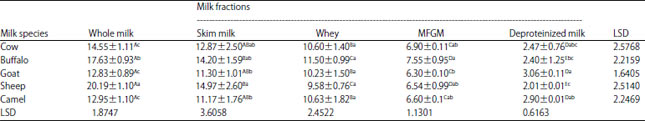
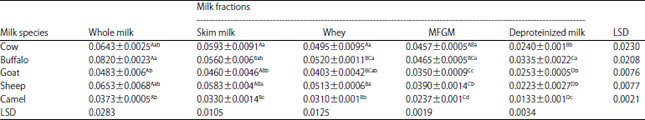
Research Article
Antioxidant Properties of Milk: Effect of Milk Species, Milk Fractions and Heat Treatments
Department of Dairy Science, Faculty of Agriculture, Cairo University, 3 El-Gamaa Street, 12613 Giza, Egypt
Mohamed Azzam
Department of Dairy Science, Faculty of Agriculture, Cairo University, 3 El-Gamaa Street, 12613 Giza, Egypt
Hany Elkashef
Department of Dairy Science, Faculty of Agriculture, Cairo University, 3 El-Gamaa Street, 12613 Giza, Egypt
LiveDNA: 20.30548
Ahmed Elhadydy
Department of Food Additives, Regional Center for Food and Feed, Agricultural Research Center, Giza, Egypt