Research Article
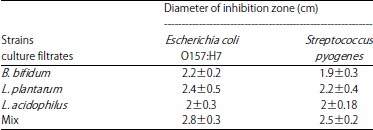
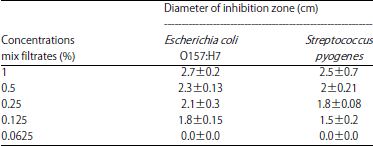
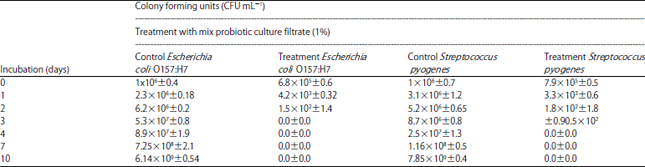
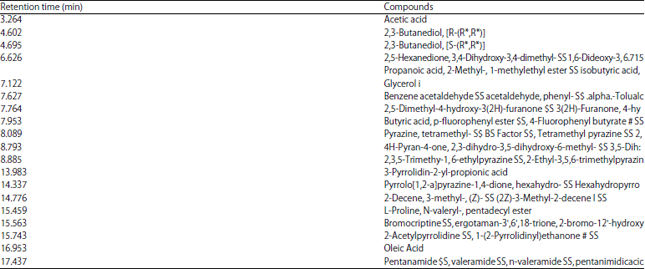
Combination of Probiotic Filtrates as Antibacterial Agent Against Selected Some Pathogenic Bacteria in Milk and Cheese
Department of Food Technology, Arid Lands and Cultivation Research Institute (ALCRI), City of Scientific Research and Technological Applications (SRTA-CITY), New Borg El-Arab City, Alexandria, Egypt
LiveDNA: 20.20321
William A. Botros
Department of Nucleic Acids Research, Genetic Engineering and Biotechnology Research Institute (GEBRI), City for Scientific Research and Technology Applications (SRTA-CITY), New Borg El-Arab City, Alexandria, Egypt
Elsayed Elsayed Hafez
Department of Plant Protection and Biomolecular Diagnosis, Arid Lands Cultivation Research Institute, City of Scientific Research and Technology Applications (SRTA-CITY), New Borg El-Arab City, Alexandria, Egypt
LiveDNA: 20.3216