Research Article
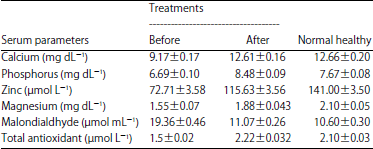
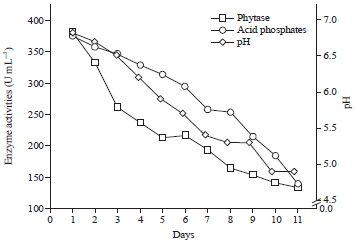
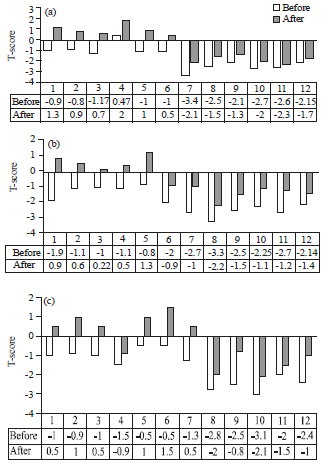
Lactobacillus reuteri can Improve BMD in Patients Having Bone Fragility
Department of Food Science and Nutrition, National Research Centre, Dokki, Giza, Egypt
Seham S. Kassem
Department of Food Science and Nutrition, National Research Centre, Dokki, Giza, Egypt
Azzat B. El-Khalek
Department of Dairy science, National Research Centre, Dokki, Giza, Egypt
Mohamed T. Fouad
Department of Dairy science, National Research Centre, Dokki, Giza, Egypt
Enas N. Danial
Department of Biochemistry, Faculty of Science Alfaisalia Campus, King Abdulaziz University, Saudi Arabia
Emtenan M. Hanafi
Departmentof Animal Reproduction and Artificial Insemination, National Research Centre, Dokki, Giza, Egypt
Maha M. Saber
Department of Complementary Medicine, National Research Centre, Dokki, Giza, Egypt
Eitedal M. Daoud
Department of Complementary Medicine, National Research Centre, Dokki, Giza, Egypt