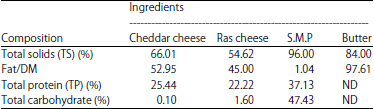
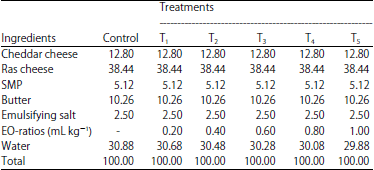
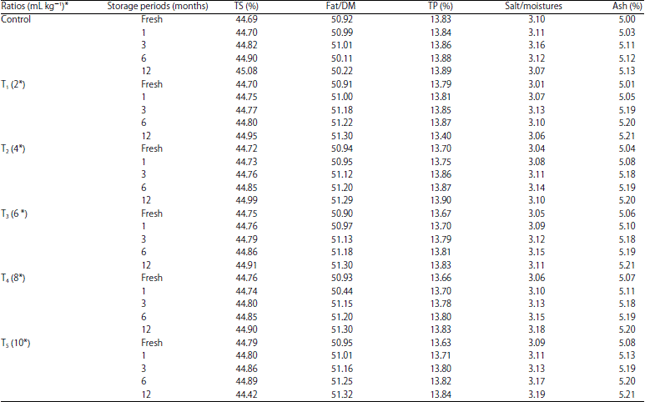
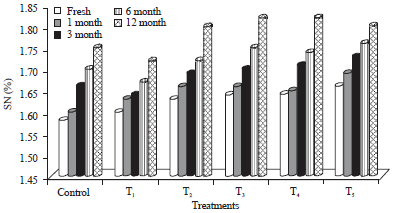
Research Article
Impact of Myrrh Essential Oil as a Highly Effective Antimicrobial Agent in Processed Cheese Spreads
Department of Dairy, National Research Centre, 33th El-Tahrir St., Cairo, Egypt
Hayam M. Abbas
Department of Dairy, National Research Centre, 33th El-Tahrir St., Cairo, Egypt
Jihan M. Kassem
Department of Dairy, National Research Centre, 33th El-Tahrir St., Cairo, Egypt
LiveDNA: 20.7763
Walid A. Gafour
Institute of Food Technology Research, Ministry of Agriculture, Giza, Egypt
A.G. Attalah
Department of Microbial Genetics, National Research Centre, 33th El th El-Tahrir St Cairo, Egypt