Research Article
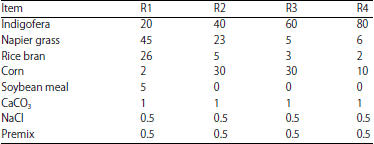
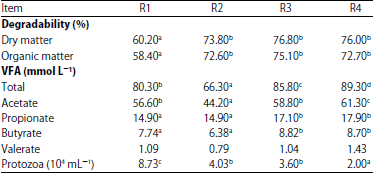
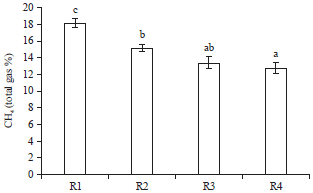
Nutritional Evaluation of Dairy Goat Rations Containing Indigofera zollingeriana by Using in vitro Rumen Fermentation Technique (RUSITEC)
Graduate School of Nutrition and Feed Science, Faculty of Animal Science, Bogor Agricultural University, Indonesia
Dewi Apri Astuti
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
Nahrowi
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
Anuraga Jayanegara
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
LiveDNA: 62.7299
Luki Abdullah
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia