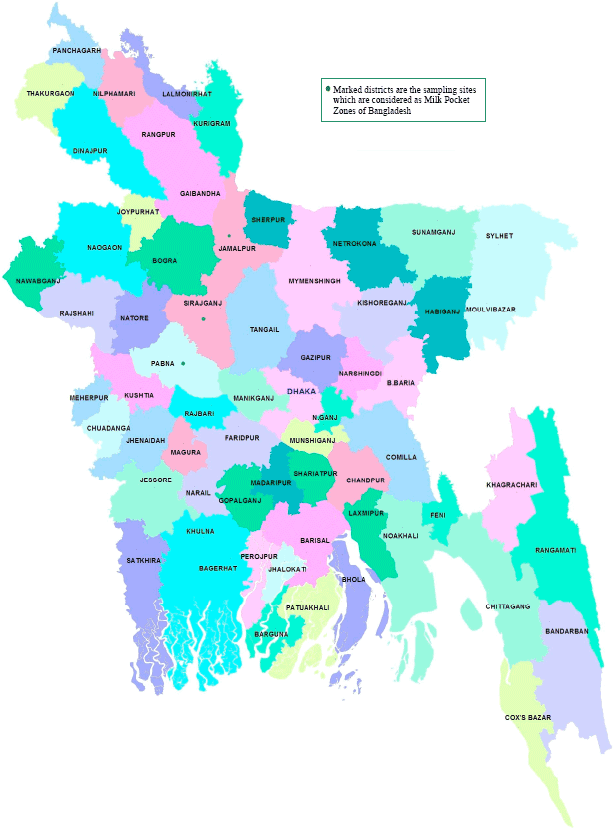
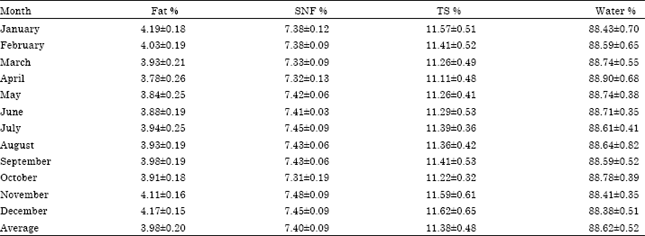
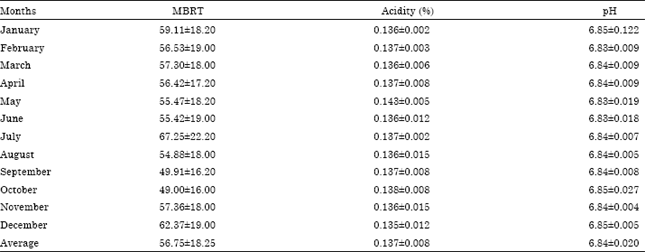
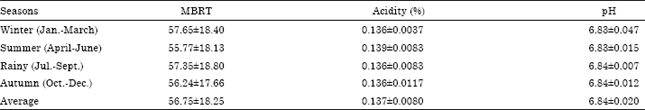
Research Article
Rapid Estimation of Quality of Raw Milk for its Suitability for Further Processing in the Dairy Industries of Bangladesh
Department of Animal Genetics and Breeding, Chittagong Veterinary and Animal Sciences University, Khulshi, Chittagong-4202, Bangladesh
Md. Manirul Islam
Department of Animal Science and Nutrition, Chittagong Veterinary and Animal Sciences University, Khulshi, Chittagong-4202, Bangladesh
M.C. Sumy
Department of Agricultural Economics and Social Sciences, Chittagong Veterinary and Animal Sciences University, Khulshi, Chittagong-4202, Bangladesh
Md. Nasim Ali Mandal
Bangladesh Milk Producer�s Co-operative Union Limited (BMPCUL), Bangladesh
Gazi Md. Noor Uddin
Department of Veterinary Disease Biology, Faculty of Health and Medical Sciences, University of Copenhagen, Stigb�jlen 4, Building 1-20, 1870 Frederiksberg C, Copenhagen, Denmark