Research Article
Weather Effect on Bacterial Mastitis in Dairy Cows
Department of Microbiology, King Faisal University, P.O. Box 1757, Al-Ahsa 31982, Kingdom of Saudi Arabia
At some point in lactation 5-10% of all dairy cows show clinical mastitis (Sci. Edu. Admn., 1980). Earlier research indicated that mastitis may be caused by approximately 90 different organisms including bacteria, fungi, proto-theca, viruses and mycoplasma (Philpot, 1978). However, Barnes and Mastitis (1984) concluded from an extensive literature review that 90-95% of all cases were due to bacterial infection. In bovine mastitis alone, over 135 bacterial genera are identified, but Streptococcus, Staphylococcus species and the coliform bacteria are the most prevalent (Barnes and Mastitis, 1984; Gonzalez and Jasper, 1990; Philpot, 1978). Gonzalez and Jasper (1990) found that environmental mastitis was predominant in most California dairy herds. However, Pankey et al. (1991) reported similar findings in Vermount and Tennessee, but in another study the contagious mastitis was prominent due to Staphylococcus aureus. Mastitis is one of the most expensive diseases affecting dairy producers in Louisiana (Shuster et al., 1991). The National Mastitis Council has estimated that mastitis losses from reduced milk production alone amounted to over $ 1 billion annually (Sci. Edu. Admn., 1980).
The long summer is characterized by intense radiant heat extending from late April or early May through mid October (Igono et al., 1992). Effects of thermal stress include reduced feed intake, reduction in metabolic rate and maintenance requirements, increased evaporative loss, increased respiration rate, change in blood hormone concentration and redistribution of total blood flow (Igono et al., 1985). In addition to causing significant reduction in milk yield, heat stress may suppress animal, thereby making it vulnerable to challenges it could handle under normal circumstances.
Although the effects of the desert climate on dairy cattle have been extensively studied, but the main focus of research remained on reproduction, housing system, coat, color and milk production (Igono et al., 1992). However, the seasonal incidence of mastitis has not been determined. This study was designed (1): to evaluate the incidence and pattern of bovine (clinical) mastitis, (2) relate the incidence of mastitis to elements of weather such as ambient temperature (t db) and the rainy days at the dairy farm and (3) to determine the bacteriological causes of clinical mastitis over a period of one year.
Meteorological Data
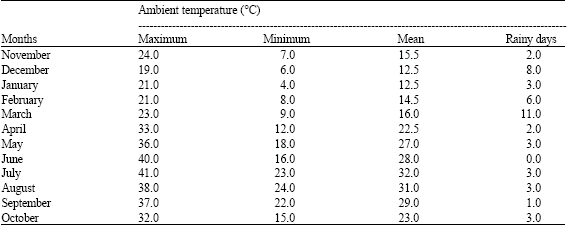
Monthly weather climate summaries (National Mastitis Council, Inc., 1990) between 1989 and 1992 were analyzed to provide a comprehensive description of the annual pattern of ambient temperature. Average maximum and minimum daily temperature and the measurable rainy days were calculated using the critical temperature estimates reported by Igono et al. (1992) as a guide, the daily dry bulb temperature (tdb) was grouped into weather periods for each day to enable in depth description of diurnal pattern of tdb. The tdb less than 21°C, 21.1-27°C and >2 7.1°C were designated as cool, warm and hot, respectively.
Dairies
Data on clinical mastitis were obtained from January, 1989 to December, 1991 from two commercial dairy farms located at Buckeye, Arizona and analyzed to generate baseline data for incidence of mastitis in relation to different seasons. The A and H Dairy Ltd., maintained an average of 789 Holstein milking cows grouped by production during 1989-1991. Cows were housed in corrals with overhead shades and earthen floors. Animals were cooled using evaporative coolers whenever, the ambient temperature was greater than 27°C.
The second dairy farm studied was the Johan Bolle Dairy (J. B. Dairy), which maintained an average of 968 Holstein milking cows grouped according to production level into five milking groups. Animals were cooled using spray cooling only at the feeding bunk in addition to spray and fan in the holding area of the milking parlor. In addition, open-sided shade structures with hanging large ventilation fans that are turned on automatically when the tdb is >30°C to manage the hot weather conditions.
Incidence of Mastitis
The rates were calculated by dividing the total mastitis incidence per day by the average number of cows per day lactating during that day.
Weather data used in the analysis were the maximum, minimum and mean dry bulb temperature and the measurable rainy days during the study period. Days in milk, until the occurrence of a new case of mastitis, were computed as the difference between the day in milk and calving date.
Milk Samples
Quarter milk samples from 90 cows with clinical mastitis in the J. and B. Dairy were taken before treatment as recommended (Brown et al., 1981). Milk samples were either frozen immediately at -5°C or placed in a portable insulated chest containing ice. Milk samples were transported to the Bacterial Systematic Laboratory ASU for examination.
Bacteriological Procedures
Refrigerated milk samples were thawed as suggested by Schalm et al. (1971) and shaken thoroughly just before removing the inoculum for plating. A sterile platinum loop was used to spread large agar plate inoculation volume (0.l mL) (Smith et al., 1982) of each milk sample into nonselective trypticase soy (BBL Microbiological Systems, Cockeysville, MD) blood agar plates containing 5% citrated bovine red blood cells (Sci. Edu. Admn., 1980). Unsuccessful attempts were done for the isolation of mycoplasma where 0.02 mL milk sample was spread on to a quarter plate of mycoplasma medium (Bacto-pPL0) agar (Difco) dehydrated as described elsewhere (Starr et al., 1979). Blood agar plates were incubated at 37°C for 24 h and examined for bacterial growth. Mycoplasma plates were observed for colonies every 48 h up to 10 days of incubation at 37°C under partial CO2 tension. Streptococci were identified using hemolytic patterns of colonies on blood agar and hydrolysis of esculin and additional biochemical tests were performed for further identification.
Staphylococci species were identified by alpha, lysin production and coagulase test and additional biochemical tests were used to differentiate from other genera such as Micrococcus. Coliforms were identified using colonial morphologic characteristics on MacConkey (BBL Microbiological Systems) agar and EMB agar (Difco), motility, citrate utilization and other biochemical tests using Enterotube II System (miniaturized multi-test system. Roche Diagnostic of Nutley, N.J.). The Corynebacterium species were identified using morphologic characteristics of colonies on nutrient agar (Difco) supplemented with Tween 80 and other recommended biochemical tests. Isolations of Moraxella bovis, Neisseria ceneria, Pasteurella haemolvtica and other aerobic and anaerobic (including Clostridium perfrinqens) gram positive bacteria were also done.
Unusual strains of organisms were suspected re-subculturing and the incubation was done at different temperature or times in an appropriate environment using thioglycollate medium when the obviously abnormal milk consistently gave no growth following the standards procedure of culturing milk, as described elsewhere (National Mastitis Council, Inc., 1990). Presumptive identification of isolates and species was carried out by following the methods of Bercrey's Manual of Systematic Bacteriology (Boone and Castenholz, 1984). Reference bacterial species were provided by the Department of Microbiology.
Statistical Analysis
The chi-square test was run to determine whether the species-specific bovine mastitis distributions were independent of season. Linear regressions were run to examine the relationship between environmental variables (temperature, dry-bulb temperature, rainfall) and the incidence of bovine mastitis. The statistical significance was set at p<0.05.
Analysis of Meteorological Data
Using critical temperatures as described by Igono et al. (1992), the seasons were defined as cold months with an average tdb <21°C, warm months with an average tdb between 21.1°C and 27°C and hot months with an average tdb >27. 1°C (Table 1).
Incidence of Mastitis
Annual incidence of bovine (clinical) mastitis for 1989, 1990 and 1991 seasons at the A and H Dairy are presented (Table 2). Within each year, there appears to be an inverse relationship between ambient temperature and the occurrence of new cases of mastitis (Table 1, 2). It was found that with increase in ambient temperature, the incidence of new cases of mastitis decreased considerably in all the three years (Fig. 1). The meteorological data from November 1991-October, 1992 are presented in Table 3. Annual incidence of new mastitis cases from November, 1991-October, 1992 in J. B. Dairy are shown in Table 4. Figure 2 shows the relationship between ambient temperature and the incidence of new cases of mastitis and clearly exhibited the inverse relationship with low incidence during hot months.
| Table 1: | Mean ambient temperature and measurable rainfall days for Buckeye, Arizona from 1989-1991 |
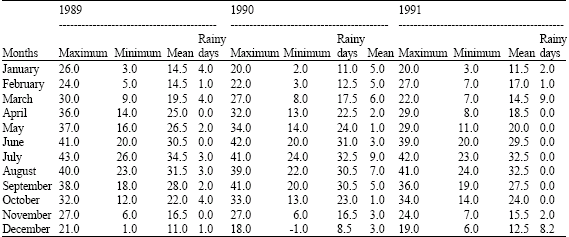 | |
| Table 2: | Annual incidence (%) of mastitis in A and H dairy in Buckeye, Arizona from January, 1989-December, 1991 |
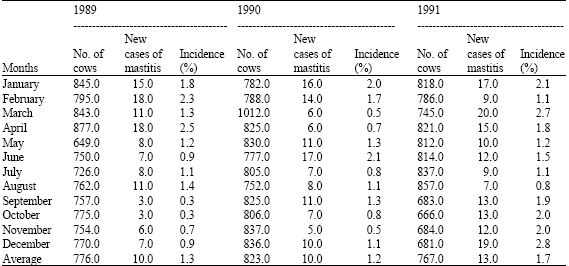 | |
| The incidence percentage was calculated by dividing the number of new mastitis cases by the average milking cows per day of the year | |
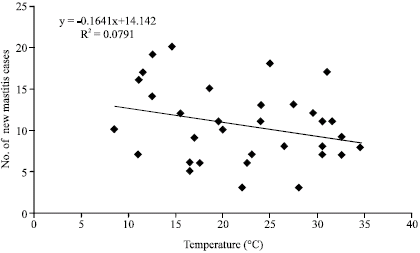 | |
| Fig. 1: | Relationship between temperature and new cases of mastitis at A and H dairy farm |
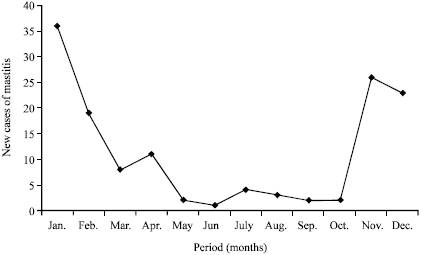 | |
| Fig. 2: | Monthly rate of new mastitis cases at J.B. Dairy |
| Table 3: | Meteorological data for Rainbow Valley, Arizona from November, 1991-October, 1992 |
 | |
| Table 4: | Incidence of mastitis in Johan Bolle Dairy in Buckeye, Arizona from November-October, 1992 |
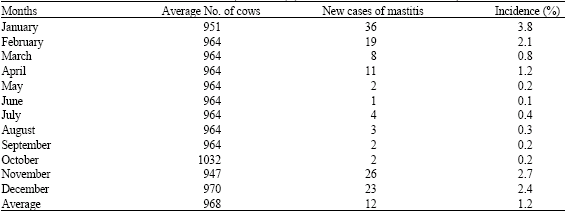 | |
| The incidence percentage was calculated by dividing the number of total new cases of mastitis by the average number of milking cows per day | |
Although the R2 value is very low (R2 = 0.079) but there is decreasing trend in new mastitis cases with increasing temperature.
Clinical Cases of Mastitis
During 1 year study, a total of 137 new cases of bovine (clinical) mastitis were detected and the most prevalent bacterial species and their incidence from 90 new mastitis cases at the J. B. Dairy were considered. Of the 90 new cases of mastitis, 21.1% were infected with coliform bacteria, 12.2 with S. haemolyticus, 11.1 due to C. bovis, 10.0 with S. agalactiae and 45.5% with other isolates.
Incidence of Mastitis
Annual incidence of new cases of mastitis based on bacterial species from November 1991 to October 1992 in the J. B. Dairy are shown in Table 5.
Table 6 represents the characterization of the environmental ambient temperature which was used to evaluate the seasonal effect on incidence of mastitis pathogens. Three main bacterial groups causing mastitis in the J. B. Dairy were most prevalent during the cold months.
| Table 5: | Annual incidence (%)6 of new cases of mastitis according to culture results in Johan Bolle Dairy from November, 1991 to October, 1992 |
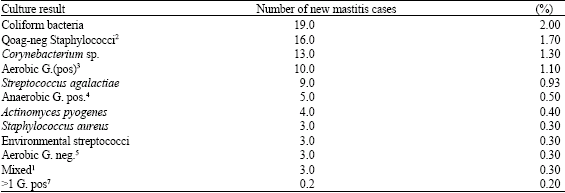 | |
| 1Both mixed gram positive and negative bacteria, 2Coagulase negative staphylococci, 3Aerobic gram positive bacteria, 4Anaerobic Gram positive bacteria, 5Aerobic gram negative bacteria, 6Computed by dividing the number of new mastitis cases by the average number of milking cows per day (968) and 7more than one gram positive bacteria | |
| Table 6: | Characterization of environmental temperature of central Arizona to evaluate the seasonal effects on the incidence of mastitis pathogens from November, 1991 to October, 1992 in Johan Bolle dairy |
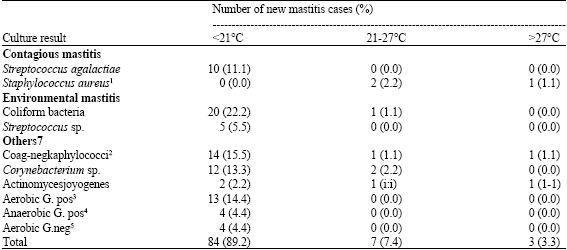 | |
| Figures in parentheses are percent of mastitis cases in relation to each group. 1(n = 3) in the isolates, 2Coagulase negative staphylococci, 3Aerobic gram positive bacteria, 4Anaerobic gram positive bacteria, 5Aerobic gram negative bacteria. 6(%) = Number of new mastitis due to certain isolates divided by the total number of isolates, 7Other environmental pathogens, 8Occured in November, December, January, February and March; 9Occured in April, May and October and 10Occured in June, July, August and September | |
| Table 7: | Mean values of chi-square test to determine if species specific bovine mastitis distribution were independent of seasons |
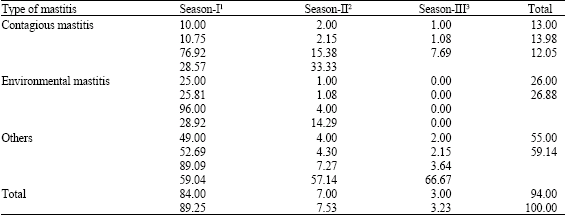 | |
| 1Cold months, 2Warm months, 3Hot months | |
Statistical Analysis
A chi-square test was used to determine if the species specific bovine mastitis distributions were independent of the season in the J. B. Dairy. Table 7 shows the relationship between seasons and the three bacterial groups which revealed a strong relationship between seasons and the bacterial groups.
The ANOVA test clearly revealed a significant effect of seasons on mastitis incidence. However, the effect of year on mastitis incidence was not significant. Similarly, the results showed significant effect of seasons on the incidence of new mastitis cases in the J. B. Dairy Farm. Overall, it was found that there existed a clear negative relationship between the new mastitis cases and the ambient temperature.
A least square linear regression analysis was run utilizing data from Table 1 and 2 which showed a significant negative relationship between the ambient temperature and the new mastitis cases (R2 = 0.11). Although the relationship was very poor, yet the data showed that the ambient temperature affects negatively the mastitis incidence. Similar analysis (R2 = 0.70) was done on data (Table 3, 4) from J. B. Dairy Farm which also showed a real negative relationship between the new mastitis cases and the ambient temperature.
Mean annual incidence of clinical mastitis at the A and H Dairy was 16.5%, which was lower than that found in some surveys on clinical mastitis (Eberhart and Buckalew, 1977; Faull et al., 1983; Gonzalez and Jasper, 1990). In the present study, the incidence of new cases of mastitis decreased with increasing ambient temperature, but increased with decreasing ambient temperature. The incidence of mastitis was higher in the cold months. However, the statistical analysis suggested significant effect of the seasons (cold months, warm months and hot months) on incidence of mastitis. The results revealed a negative relationship between the seasons and the mastitis incidence.
There were 14.4% annual incidence of clinical mastitis at J. B. Dairy and were lower than those found by other surveys on clinical mastitis (Gonzalez and Jasper, 1990). The relationship between ambient temperature and the mastitis incidence was similar to that obtained for A and H Dairy. It was observed that there existed a negative relationship between the ambient temperature and the incidence of mastitis and these findings were identical to those obtained for A and H Dairy.
Analysis of milk samples from new mastitis cases revealed no culture negative samples which were not in agreement with the findings of Eberhart and Buckalew (1977) and Erskine and Eberhart (1991) where at least 19% accounted for culture negative samples. A possible explanation is that using large inoculum milk samples to streak a non differential medium could have helped to screen low bacterial population in milk samples. Present study suggests that standard 0.0l mL inocula might not be sensitive enough to detect small bacterial populations in a milk sample.
Bacteriologic examination of milk samples from new cases of mastitis indicated that environmental mastitis pathogens were causing serious clinical mastitis in J. B. Dairy. Environmental and contagious mastitis pathogens were identified in 43.3% of all the clinical mastitis cases. However, coliform bacteria were the main isolated mastitis pathogens and accounted for 21.1% of the total clinical mastitis cases. Coliform incidence was higher than that found by Pankey et al. (1991) but lower than those reported by Eberhart and Bucklew (1977) in Reading, England. Serratia marcescens was reported as the main cause of clinical mastitis (Ruegg et al., 1992). Other researchers indicated that the that the main mastitis pathogens were coagulase negative Staphylococci (Bushnell et al., 1980; Erskine and Eberhart, 1991) whereas veterinarians in Denmark reported that the main mastitis pathogen was P. indolicus (Madsen et al., 1990).
In this study, S. aureus accounted for 3.3% of new mastitis cases, which differs from those stated by Trinidad et al. (1990) who reported much higher prevalence (37.1%). The value of 3.3% in this study is higher than that reported by Pankey et al. (1991). During this study, S. agalactiae were diagnosed in 10% of the new mastitis cases, which is higher than that found in clinical mastitis in Tucson, Arizona (Bushnell et al., 1980) but lower than that in Pennsylvania stated by Erskine and Eberhart (1991) who reported that S. agalactiae accounted for 25% of the mastitis cases.
The E. coli in this study was the most abundant coliform bacteria from new cases of mastitis and is in agreement with those described by Faull et al. (1983) and Gonzales and Jasper (1990), but differed from the findings of Eberhart and Buckalew (1977) where K. pneumoniae was more frequently isolated. However, E. coli mastitis is an important cause of morbidity and is one of the most serious challenges facing dairy practitioners (Lohuis et al., 1990). Even if the animal survives, it is difficult for the animal to return to normal lactation (Shuster et al., 1991).
Overall, this study showed that the highest clinical mastitis incidence for almost all the bacterial isolates occurred in cold months. The peak of coliform bacteria was when the tdb was less than 21°C especially following the rainy and muddy periods. In Kentucky, Harmon et al. (1992) showed that the mastitis pathogens tended to be highest in spring and summer months. They also reported that streptococcal mastitis pathogens were higher than coliform pathogens and were associated with periods of elevated ambient temperature.
Identical conclusions were drawn for the seasonal influence on clinical mastitis. Similarly, Batra et al. (1977) did not find any seasonal relationship associated with clinical mastitis. On the other hand, in Helsinki, Finland; Pyorala et al. (1992) reported that lactating cows were more often affected by bacterial mastitis in winter. Faull et al. (1983) observed in England that E. coli clinical mastitis was higher in summer months than cold months of the year. This variability in findings between the present study and their study could be related to the occurrence of clod months being November, December and January in Great Britain and from November through March in this experimental conditions.
The researchers thank Marilyn Jean Bloom and Dr. Ali Jumaah for technical assistance, Mr. John M. Bolle for his cooperation to work at his farm, A. and H. Dairy people for their assistance in collecting useful data, office of Climatology, for required climate information and the animal care unit at ASU for their sincere cooperation during the study period.