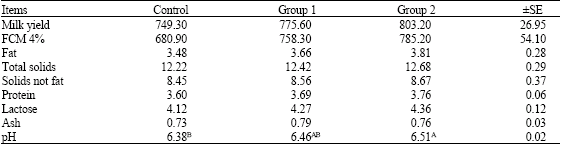
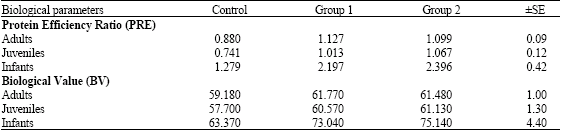
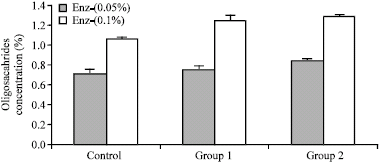
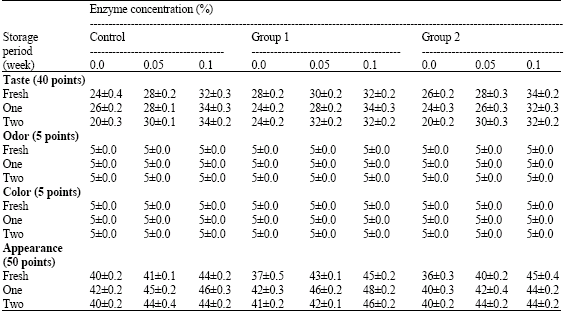
Research Article
Evaluation of Goat Milk Containing Galactooligosaccharides After Supplementing the Ration with Amino Acids
Department of Dairy, National Research Center, Dokki, Cairo, Egypt
S.M. Kholif
Department of Dairy, National Research Center, Dokki, Cairo, Egypt
A.M. Kholif
Department of Dairy, National Research Center, Dokki, Cairo, Egypt