Short Communication
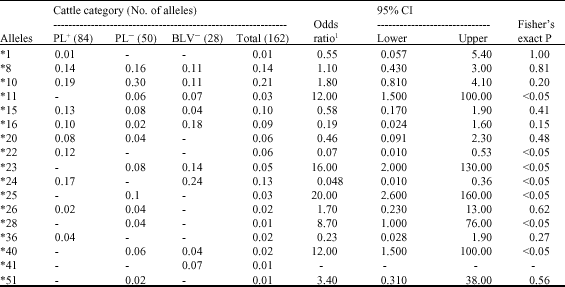
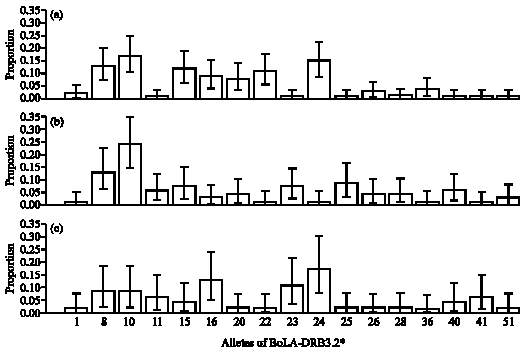
Association of BoLA-DRB3.2 Alleles with Resistance and Susceptibility to Persistent Lymphocytosis in BLV Infected Cattle in Argentina
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina
K. Suzuki
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina
M.G. Echeverria
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina
M.S. Serena
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina
G.E. Metz
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina
E.T. Gonzalez
Catedra de Virologia, Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata (FCV-UNLP), La Plata, 1900, Argentina