Research Article
Prevalence and Significance of Staphylococcus aureus and Enterobacteriaceae species in Selected Dairy Products and Handlers
Department of Animal Hygiene and Zoonoses, Milk Hygiene and Control, Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt
Jehan Ismail
Department of Food Hygiene, Milk Hygiene and Control, Faculty of Veterinary Medicine, Suez Canal University, Ismailia, Egypt









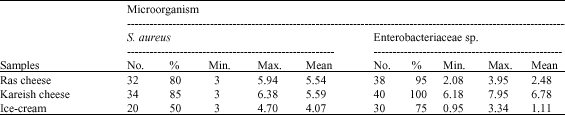
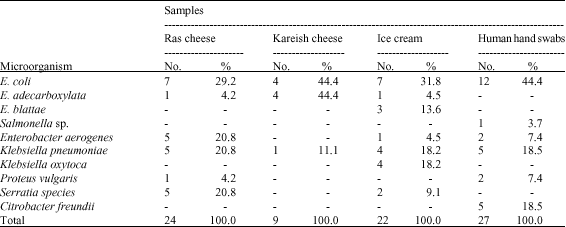
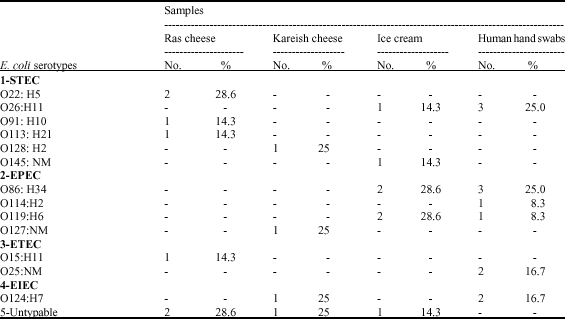

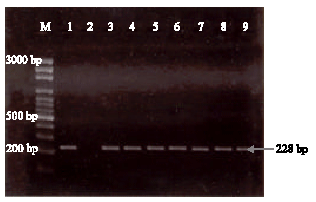
gaikwad sunil manikrao Reply
this is very good research work and is very essential work done by the author(s). best wishes for future investigatiion