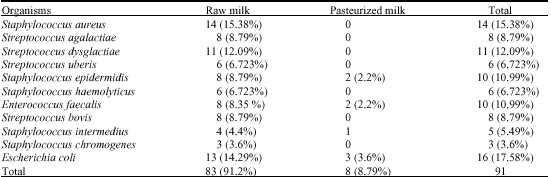
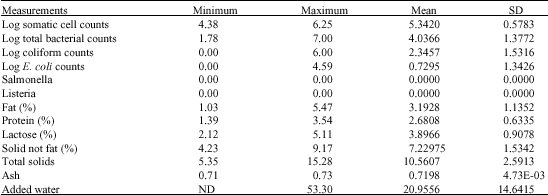
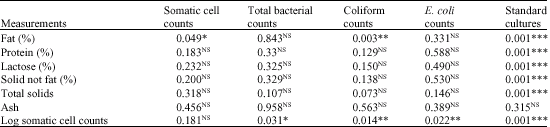
Research Article
Comparison of Chemical Composition and Microbial Profile of Raw and Pasteurized Milk of the Western Cape, South Africa
Department of Dairy Production, Faculty of Animal Production, University of Khartoum, P.O. Box 32, Postal Code 13314, Khartoum North, Sudan
Voughon Gabriechise
Department of Medical Microbiology, Faculty of Natural Science, University of the Western Cape, Private Bag X17, Bellville, 7530, South Africa
Q. Johnson
Department of Medical Microbiology, Faculty of Natural Science, University of the Western Cape, Private Bag X17, Bellville, 7530, South Africa