Research Article
Studies on the Microbiology and Nutritional Qualities of a Nigerian Fermented Milk Product (Nono)
University of Agriculture, Abia State, Nigeria
M.J. Ikenebomeh
University of Benin, Benin City, Edo State, Nigeria
Introduction
Nono is a Nigerian milk food similar to Yoghurt and other fermented milk products that is traditionally produced and consumed particularly by the Hausa and Fulanis of Nothern Nigeria. It is the product of spontaneous fermentation caused by the introduction of starter culture into fresh, raw, unskimmed milk (Manshamu). Nono is produced locally in contaminated environment by hand-milking of cows into large calabashes (Kworia), that is, crude cultured with subsequent skimming to remove the surface cream that becomes the Fulanis butter which is a by -product of the preparation. Nono can be consumed on its own or used in the preparation of milk cereal based food known as Fura-Nono (Eka and Ohaba, 1997). Although acclaimed traditionally to have better nutritional values than the unfermented milk, Nono is basically kept unrefrigerated and its acceptability depends on the texture, flavour and taste which are in turn dependent upon the inherent microbial constituents. Due to the gross unhygienic method of preparation and handling, Nono has been claimed to be highly contaminated with such organisms like Staphylococcus aureus, Escherichia coli, Bacillus cereus and Pseudomonas aeruginosa (Atanda and Ikenebomeh, 1988).
During milk production, microbial contamination generally occurs from three main sources: from within the udder, from the exterior of the udder and from the surface of milk handling and storage equipment (Feresu and Muzondo, 1990). Equally important are the temperature and length of time of storage, which allow microbial contaminants to multiply and increase in numbers. All these factors will influence the total bacterial count or Standard Plate Count (SPC) and the types of bacteria present in bulk raw milk.
Raw milk is milk in its natural (unpasteurized) state (Freitas and Malcata, 2000). Milk can be considered as containing three basic components: water, fats and Non-fatty Solids (NFS). The organic matter in the non-fatty portion consists mainly of the proteins casein, albumin and globulin; lactose and lactic acids. Milk essentially is 88% water and 12% solids. Of these solids, 4% are fat, 3.5% are protein, 4% are lactose (milk sugar) and 0.5% are minerals (Freitas and Malcata, 2000). Public health authorities advocate pasteurization of fresh milk to destroy any disease-producing bacteria that may be present especially mastitis organisms. Notably among these are Streptococcus agalactiae and S. uberis (Grippon, 1997).
However, health analysts claim that it destroys essential nutrients. Contaminated raw milk can be a source of harmful bacteria, such as those that cause undulant fever, dysentery, salmonellosis and tuberculosis. Certified milk, obtained from healthy cows is unpasteurized milk with a bacteria count below a specified standard, but it still can contain significant numbers of disease-producing organisms (Feresu and Nyati, 1990). Hence, consumption of raw milk should be discouraged.
The objective of the present study is to determine the microbilogical and Nutritional changes that take place During the traditional fermentation of Raw mlik and the possible reasons that make the fulani’s in Nigeria take the fermented product (Nono) more than raw milk.
Materials and Methods
The present study was conducted in 2004 in Benin-city, Edo state, Nigeria. Sterile universal bottles were used in collecting samples of raw milk from lactating cows with the assistance of Fulani cattle rearers at Enyean and Aduwawa in Benin-City, Edo State. The samples were transferred into coolers containing ice-blocks and quickly taken to the laboratory, kept in the refrigerator until they were used. The raw milk sample was aseptically transferred into sterile 1000 mL beaker, covered with aluminum foil and left on the bench to be fermented by the indigenous organisms present in the milk. The sample was plated on the prepared media at zero hour at the beginning of the fermentation experiment. The sample was plated unto Nutrient Agar, McConkey Agar, Yeast extract agar and Malt extract agarat zero hour and at every two-hour interval till the fermentation was over after 12 h.
Physico-chemical Analyses
The temperature and pH of the sample were determined using single electrode pH meter while the Titratable Acidity (TA) was determined for the fresh and fermented milk. The method of titration was that of Beran et al using phenolphthalein indicator by dispensing 20 mL of 0.1 N NaOH solution into a 250 mL conical flask. Two drops of the indicator were added to the solution in the flask. The milk sample was then titrated against the solution in the flask until a pink consistency was obtained. The raw milk quality was determined by Methylene blue test as described by Uraih and Izuagbe (1990).
Proximate Analysis
This was carried out to determine the nutritional status of the milk samples. The moisture content of the samples was determined based on the principle of drying to constant weight as described by Osborne and Voogt (1978). The ash content was determined then by dry-washing in a Muffle Furnace at 500°C according to Bell and Favier (1980) and Osborne and Voogt (1978).
Cholesterol values were determined according to Varley (1989) and Wonton (1964). The same was applied to the determination of the total protein, albumin, glucose and High Density Lipoprotein (HDL) while low density lipoprotein values were determined by calculation using the formula:
Where TC = Total Cholesterol value; TG = Triglyceride; HDL = High Density Lipoprotein; LDL = Low Density Lipoprotein. Standard plate count values were determined according to Fawole and Oso (1995).
The resultant bacterial colonies that resulted after incubation at 37°C for 24 h were characterized and identified with the criteria of Ogbulie et al. (1998) and Buchman and Gibbons (1974) while the fungal growths that resulted after 14 days incubation at 22°C were identified according to Barnett (1972) and Jawetz et al. (1995).
A total of one hundred and twenty organisms (120) were isolated from milk samples in the course of fermentation and can be separated into eight genera and species. These include Staphylococcus aureus, Bacillus cereus, Pseudomonas aeruginosa, Escherichia coli, Enterobacter aerogenes, Lactobacillus lactis, Geotricum candidum and Saccharomyces cerevisiae. Milk quality test showed that the milk sample was decolourised within the first thirty minutes of incubation while a total of 2.1x106 colony forming units per ml were obtained from the serial dilution cell count. Results of the nutritional status of the milk samples at different stages of fermentation, percentage nutritional status and titration parameters are shown in Table 1-3, respectively.
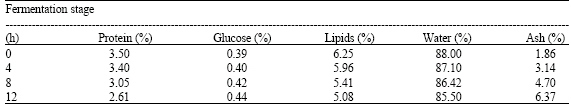
| Table 1: | Percentage nutritional status of milk at different stages of fermentation |
 | |
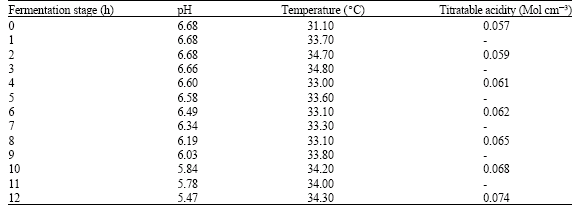
| Table 2: | pH, temperature and titratable acidity during fermentation of milk to produce Nono |
 | |
| -; Not measured | |
| Table 3: | Nutritional status of milk samples during fermentation |
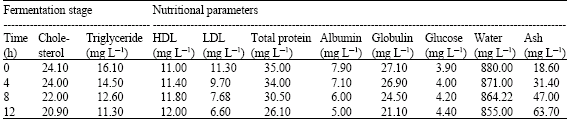 | |
| HDL = High Density Lipoprotein, LDL = Low Density Lipoprotein, Mg/l = Milligram per mL-1 | |
The persistence of all but S. cerevisiae the pH of 5.47 is an indication that Nono should be pasteurized since the pathogenic organisms were not destroyed at that pH. The occurrence of B. cereus and G. candidum in the milk sample was due to contamination of the milk and the cow’s udder with cow dung, soil and organic materials from the cow's environment since the two organisms are strictly saprophytes (Jawetz et al., 1995). Therefore, good hygienic practices like cleaning of the cow's udder with sanitizing solutions (e.g., Spray, wet towels or dip) especially during milking will reduce or totally eliminate such microbes from the milk (Fox and McSweeny, 1998; Sanchez et al., 1993).
The result also showed a marked decrease of 13.28 and 29.81% in the final values of the milk cholesterol and triglyceride, respectively. This was due to microbial breakdown of these components during the fermentation. The decrease in value recorded is of good nutritional importance as high cholesterol value of dairy foods (e.g., milk) has been implicated in recent time as a major contributor to most heart-associated problems such as cholesterosis, liver cirrhosis and cholestasis especially among the aged (Freitas and Malcata, 2000). Consequently, the consumption of pasteurized fermented milk products will stem the tide in the occurrence of these problems.
The recorded pH of 6.68 for fresh milk indicated that milk is a suitable growth medium for heterogenous microbes since most of them grow best at a physiological pH of 6.80 (Uraih and Izuagbe, 1990). The drop in the final pH value was due to the activities of L. lactis; however, the initial pH of 6.68 must have contributed greatly to the high microbial count of 2.1x106 cfu mL-1 obtained from the plate count. This number will obviously increase if the milk is left at room temperature. As a follow up to that, raw milk should be refrigerated at a temperature of 4±1°C for not more than 10 days, otherwise, the milk will deteriorate by the activities of psychrophiles like P. aeruginosa found in the milk (Fox and McSweeny, 1998).
The increase in the final glucose content of the fermented milk was due to microbial breakdown of milk carbohydrate (lactose) to glucose and galactose. This means that fermented milk products are faster in energy production in the body than unfermented milk which has only lactose (Emmons et al., 1990). This may be the underlying reason that make the Fulanis take Nono regularly. Interestingly, the slight acidic nature of the fermented milk is of good medicinal value to health as the implantation of the lactic acid in the intestines reportedly replaces the putrefying microflora therein whose metabolites have been considered to be responsible for various human ailments in the Balkan countries that finally led to premature death of the inhabitants (Freitas and Malcata, 2000).
Results obtained showed an increased yield in the HDL and a low yield in LDL. The HDL transports cholesterol from the arteries to the blood (Maisto et al., 1999), while the LDL does the reverse. Thus, more cholesterol will be transported to the blood by HDL to be used in energy generation in the cells when there is a drop in the level of triglycerides. This will consequently lead to a drop in the level of cholesterol in the body. There were marked decrease in the final protein, moisture and ash values of the fermented milk indicating that the product has lower protein value than fresh milk. Thus, Nono does not contribute more protein than milk to the body -a major finding from the research work. Conclusively, the Nono examined contained microorganisms of health importance while the nutritional status varied with fermentation. Its unpasteurised consumption is not advocated.
Result showed that fermentation did Not eliminate pathogenic organisms from The final product. Thus, it is dangerous to Consume the “nono” without Pasteurization or boiling in the traditional setting. Also, there were decrease in the total protein, triglyceride, water and Cholesterol (LdL) values of the fermented milk compared with the raw milk while The glucose, ash (minerals) and HDL values of the fermented milk increased above the values of the raw mlik, respectively. This means that Nono is a good source of energy to the consumes as seen from the greater glucose content in the fermented product, but with lower protein value than the raw milk. The higher glucose value (due to the breakdown of lactose in milk to glucose and galatose by some of the organisms in the milk) may be the reason that makes the fulanis in Nigeria to G for the Nono as part of their daily food than the raw milk. Furthermore, the consumption of Nono helps in elimination of heart problems associated with high cholesterol level in the blood.
safiyan mohammed Reply
good day,am safiyanu from nigeria. please i need method and matirial on nutritional study of fresh and fermated milk. THANK YOU