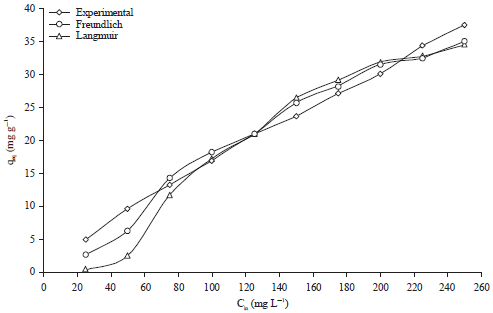
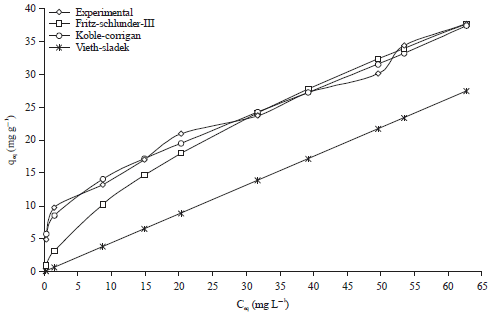
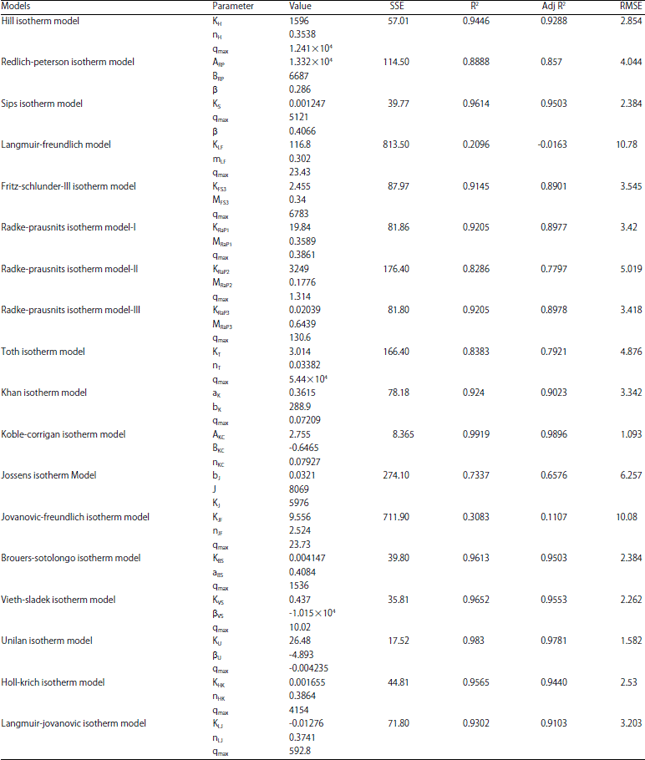
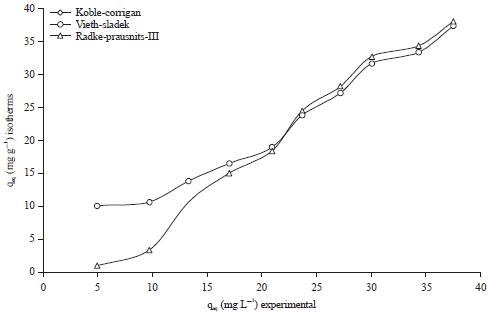
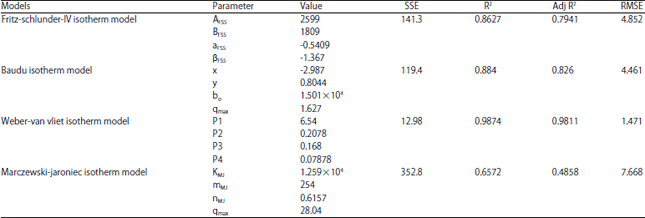
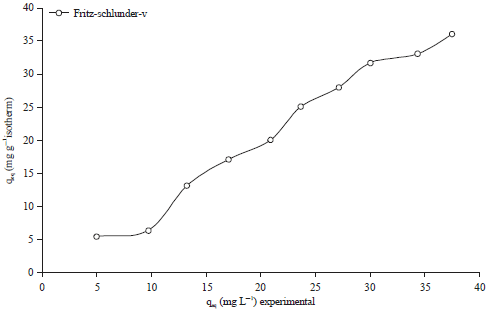
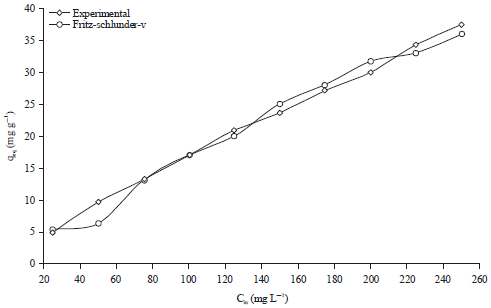
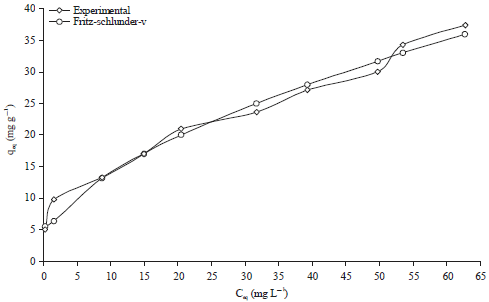Research Article
Adsorption of Chromium Using Blue Green Algae-Modeling and Application of Various Isotherms
Department of Chemical Engineering, Faculty of Engineering and Technology, Bioprocess Laboratory, Annamalai University, 608002 Annamalai Nagar, Tamil Nadu, India
Dhanasekaran Subramaniam
Department of Chemical Engineering, Faculty of Engineering and Technology, Mass Transfer Laboratory, Annamalai University, 608002 Annamalai Nagar, Tamil Nadu, India
LiveDNA: 91.22355