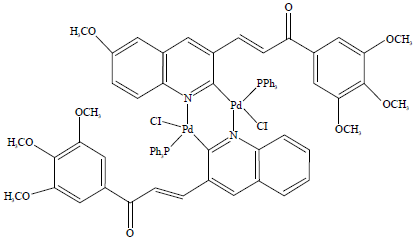
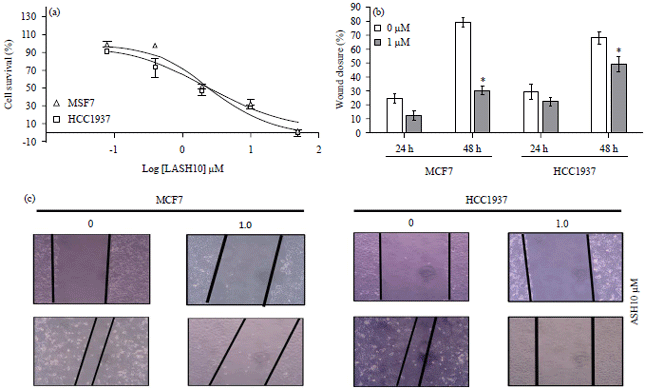
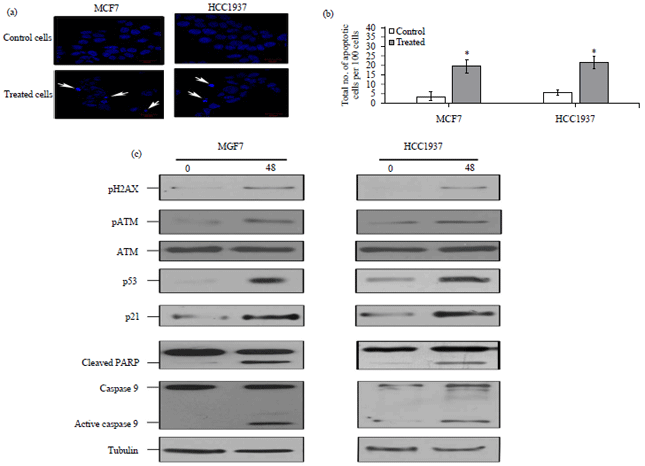
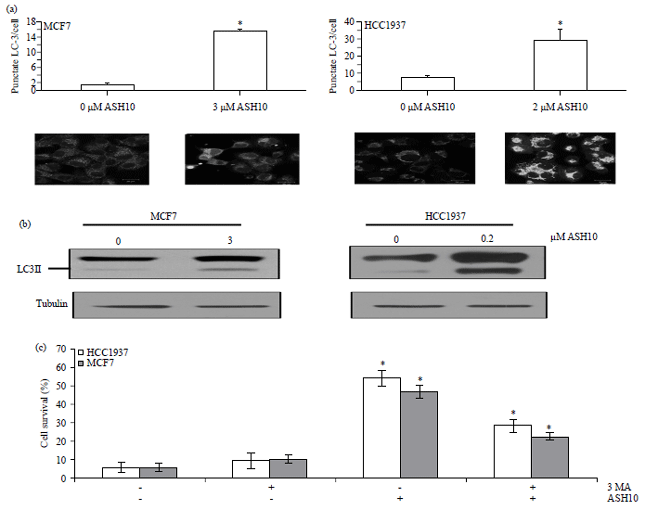
Research Article
Newly Synthesized Palladium (II) Complex ASH10 Induces Apoptosis and Autophagy in Breast Cancer Cells
Department of Clinical Laboratory Sciences, Faculty of Science, University of Jordan, Amman, Jordan
Abdel-Sattar S. Hamad Elgazwy
Department of Chemistry, Faculty of Science, Ain Shams University, Abbassia, 11566 Cairo, Egypt
Saeb Aliwaini
Department of Biological Sciences and Biotechnology, Faculty of Sciences, Islamic University of Gaza, Gaza 108, Palestine
LiveDNA: 970.15194