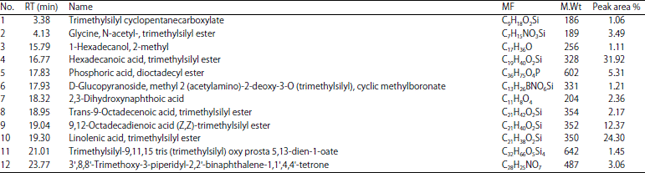
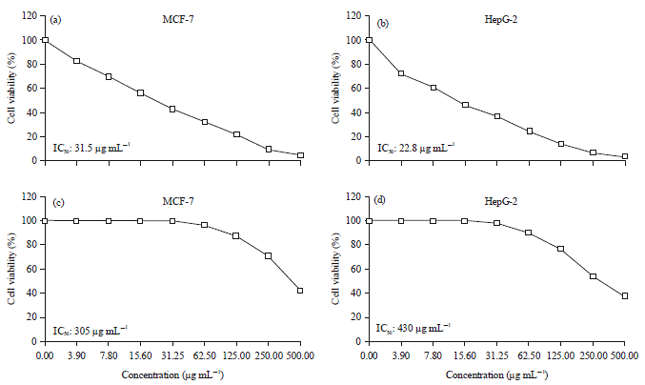
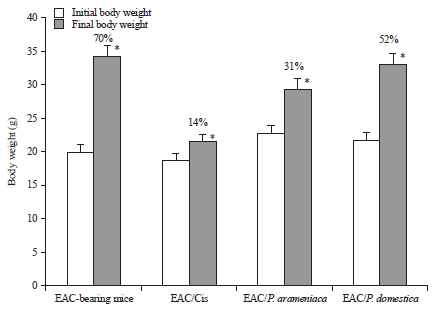
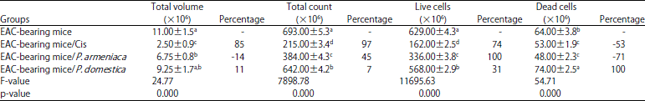
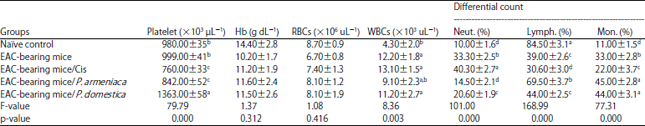
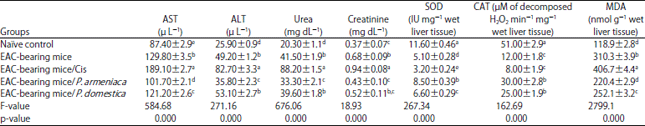
Research Article
Cancer-fighting Phytochemicals of Prunus armeniaca and Prunus domestica Seeds Extracts
Department of Zoology, Faculty of Science, Tanta University, Tanta, Egypt
LiveDNA: 20.2330
Karim Samy El-Said
Division of Biochemistry, Department of Chemistry, Faculty of Science, Tanta University, Tanta, Egypt
Adel Abd Al Hameed Albagoury
Division of Nutrition and Food Science, Department of Home Economics, Faculty of Specific Education, Tanta University, Tanta, Egypt
Violet Sobhy Awad
Division of Nutrition and Food Science, Department of Home Economics, Faculty of Specific Education, Tanta University, Tanta, Egypt
Laila Hamada Attia
Division of Nutrition and Food Science, Department of Home Economics, Faculty of Specific Education, Tanta University, Tanta, Egypt