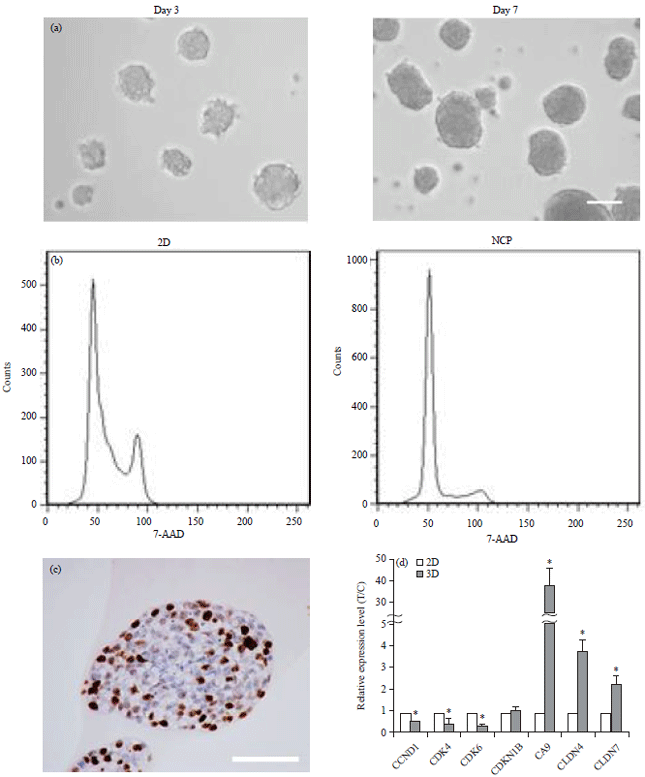
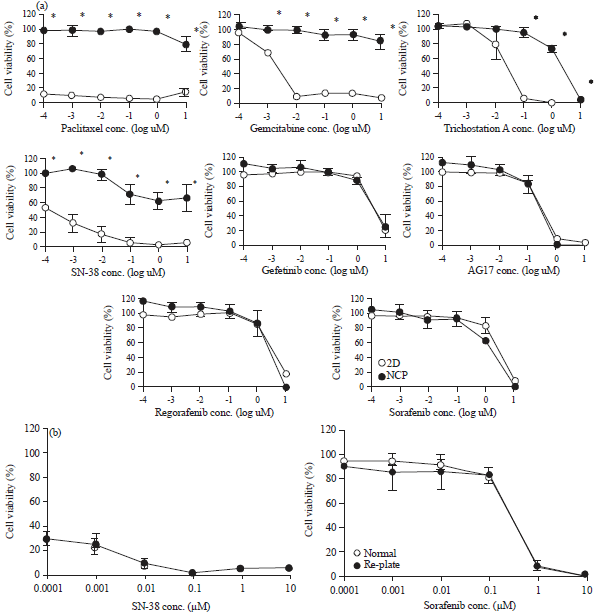
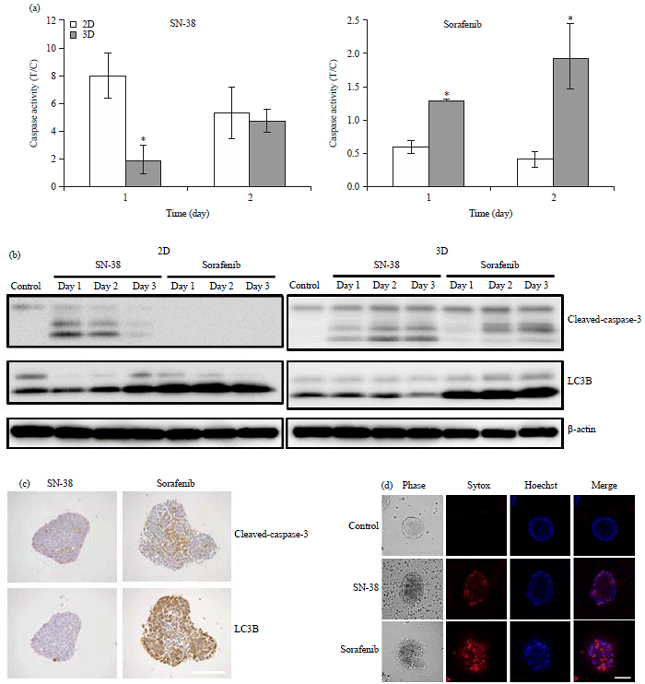
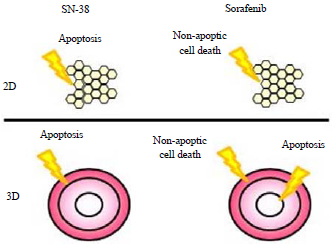
Research Article
Analysis of Sensitivity and Cell Death Pathways Mediated by Anti-cancer Drugs Using Three-dimensional Culture System
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
Hajime Higuchi
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
LiveDNA: 81.22513
Ayano- Kabashima-Niibe
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
Gen Sakai
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
Yasuo Hamamoto
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
Hiromasa Takaishi
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan
Takanori Kanai
Department of Internal Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjyuku-ku, 160-8582 Tokyo, Japan