Research Article
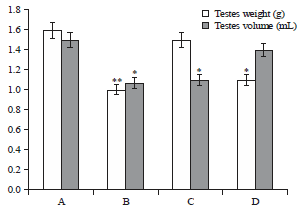
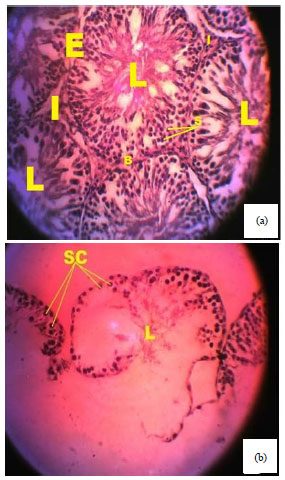
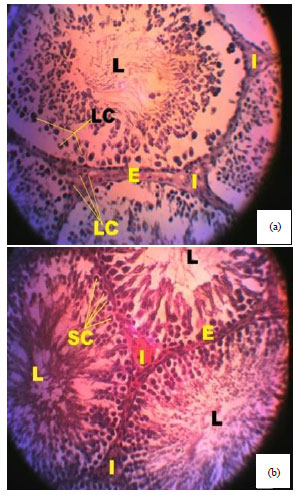
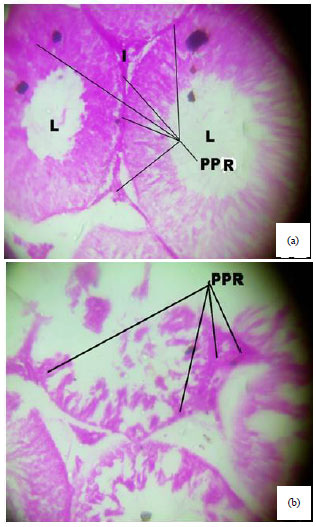
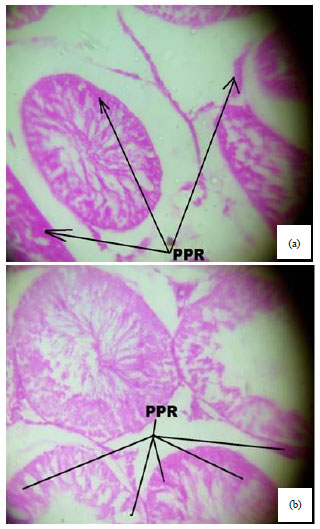
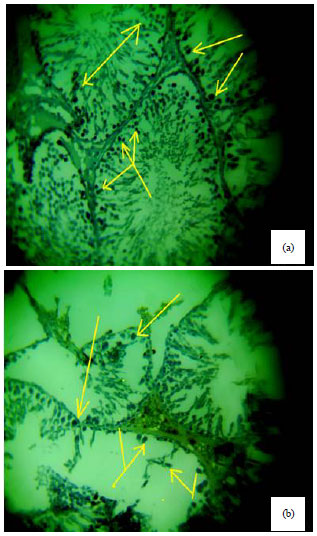
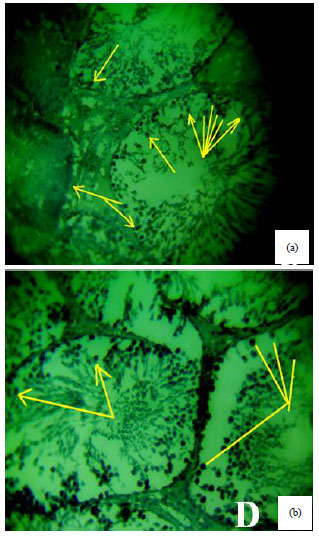
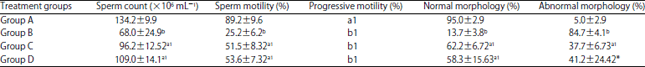
Spermiographic, 2 and 3-dimensional Quantitative Analysis of Testicular Tissues of Rat Submitted to Citrus paradisi Waste Extract and Cisplatin-induced Cytotoxicity
Department of Anatomy, Faculty of Basic Medical Sciences, Federal University Ndufu Alike Ikwo, Ebonyi State, Nigeria
E.N. Obikili
Department of Anatomy, Faculty of Basic Medical Sciences, University of Nigeria, Enugu Campus, Nigeria
G.E. Anyanwu
Department of Anatomy, Faculty of Basic Medical Sciences, University of Nigeria, Enugu Campus, Nigeria
E.A. Esom
Department of Anatomy, Faculty of Basic Medical Sciences, University of Nigeria, Enugu Campus, Nigeria











Elijah B. Kelvin Reply
Nice one