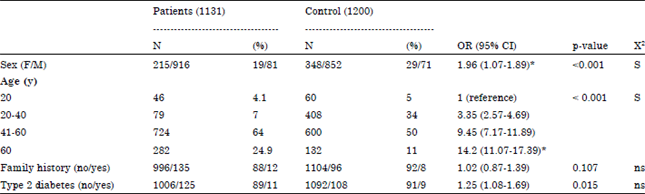
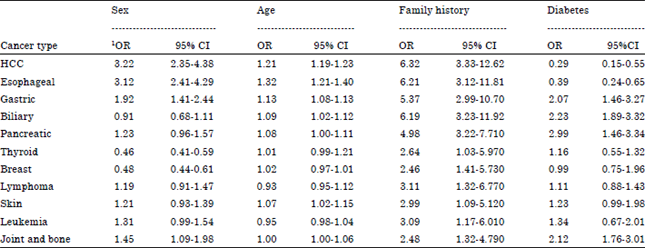
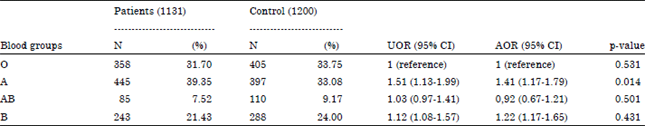
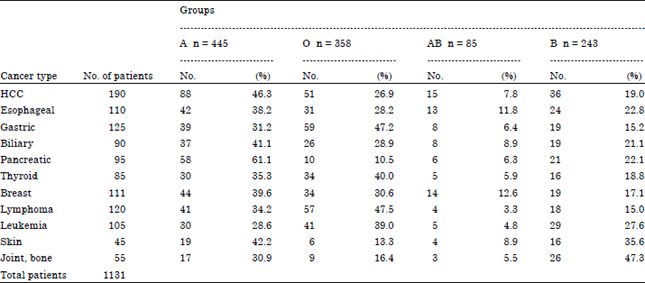
Research Article
ABO Blood Group and Risk of Malignancies in Egyptians
Department of Clinical Pathology,Faculty of Medicine, Mansoura University, Egypt
Mohammed Amin Mohammed
Department of Clinical Pathology,Faculty of Medicine, Mansoura University, Egypt
Rokia Anwar
Department of Clinical Pathology,Faculty of Medicine, Mansoura University, Egypt
M.E. Elzafrany
Oncology Centre,Faculty of Medicine, Mansoura University, Egypt
Nesreen Moustafa Omar
Oncology Centre,Faculty of Medicine, Mansoura University, Egypt