Research Article
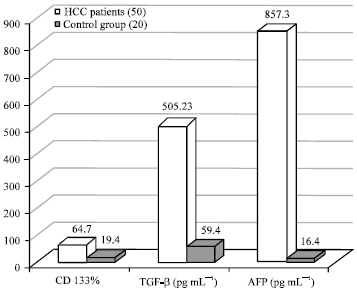
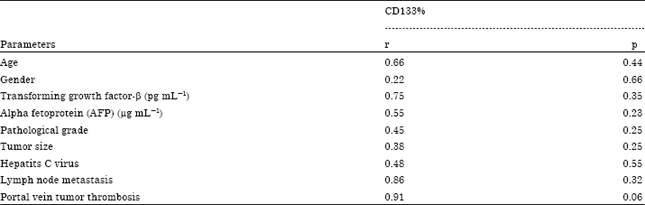
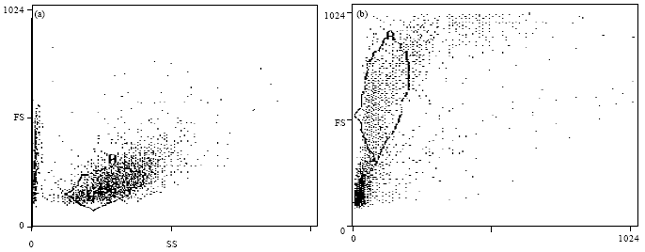
Regulation of Cancer Stem Cell Marker (CD133) by Transforming Growth Factor Beta in Hepatocellular Carcinoma
Department of Clinical Pathology,Faculty of Medicine, Mansoura University, Egypt
Tawfik R. Elkhodary
Oncology Center Mansoura University,Faculty of Medicine, Mansoura University, Egypt
Rokia Anwar
Department of Internal Medicine, Faculty of Medicine, Mansoura University, Egypt
Maha Ragab Habeeb
Department of Internal Medicine, Faculty of Medicine, Mansoura University, Egypt
Mohammed Amin Mohammed
Department of Internal Medicine, Faculty of Medicine, Mansoura University, Egypt