Research Article
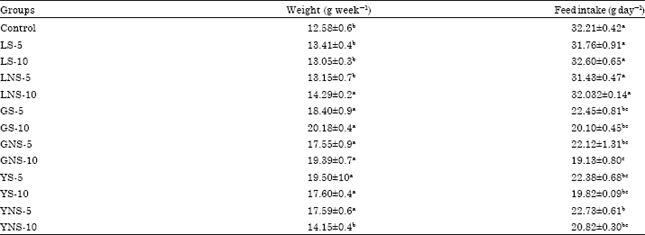
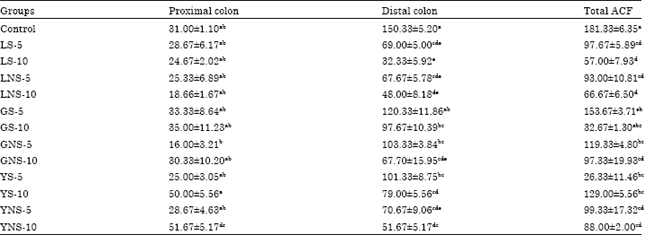
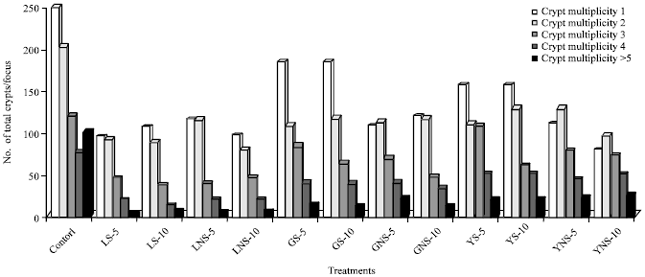
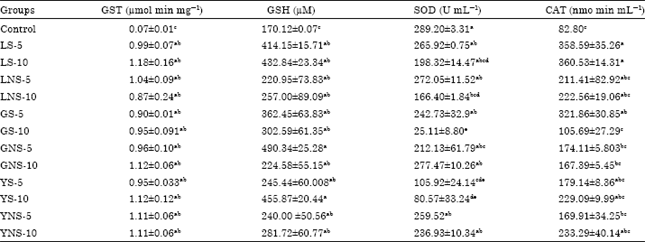
Inhibitory Effect of Lentils, Green Split and Yellow Peas (Sprouted and Non-sprouted) on Azoxymethane-induced Aberrant Crypt Foci in Fisher 344 Male Rats
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
R.L. Miller-Cebert
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
L. Aboagye
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
L. Dalrymple
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
J. Boateng
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
L. Shackelford
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
L.T. Walker
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America
M. Verghese
Department of Food and Animal Sciences, Nutrition Biochemistry and Carcinogenesis Laboratory, Alabama A and M University, Normal, Al, 35762, United State of America